GRADE
11
Chemistry SCH3U
TABLE OF CONTENTS
Matter, Chemical Bonding, and Naming
Background: Elements
Which of the following is not a metal?
Solution
Silicon (Si) is a non-metal
Background: Atomic Numbers
Determine the number of neutrons in the isotope of Copper:
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
neutrons
Hint Unavailable
Background: Substances
A compound is defined as
Solution
Compounds are molecules that consist of two or more different elements
Bohr Nuclear Model
The following three terms are equivalent:
Orbit, Shell, Energy Level
Electron Transitions
In an atom, energy is released in the form of light in certain wavelengths when:
Electrons on Shells (Energy Levels)
The maximum number of electrons on the third energy level is:
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
electrons
Hint Unavailable
The maximum number of electrons on an energy level equals 2(n)2.
2(3)2 = 18.
2 electrons in the first, 8 in the second, and actually 18 in the third.
2(3)2 = 18.
2 electrons in the first, 8 in the second, and actually 18 in the third.
Periodic Trends
Atomic radius increases across a period because of the greater atomic number.
Solution
Atomic radius decreases across a period (row) because more protons attracts more electrons and squeezes the radius smaller. The greater positive nuclear charge of protons attracts the greater number of negative valence electrons.
Which element will have the largest atomic radius?
Solution
Atomic radius increases down a group, and from right-to-left across a period. The lower-left elements have the largest atomic radius (size).
Periodic Trends: Atomic Radius of Anions
Predict the reason neutral elemental oxygen (O) will have a larger or smaller atomic radius compared to its oxide (O2-) ion.
Solution
Most people think the greater valence electron charge on the oxide (O2-) ion causes a stronger attraction with the positive nuclear charge resulting in a smaller radius - but this is not correct.
Negative ions actually have a larger atomic radius than the neutral atom because the extra electrons cause greater electron-electron repulsions, increasing the size of the electron cloud.
(Positive ions have a lower atomic radius then the elemental state)
Negative ions actually have a larger atomic radius than the neutral atom because the extra electrons cause greater electron-electron repulsions, increasing the size of the electron cloud.
(Positive ions have a lower atomic radius then the elemental state)
Periodic Trends: Atomic Radius of Cations
The atomic radius of a cation is always smaller than the original neutral element.
Solution
Because removing an electron decreases some of the electron-electron repulsions in a neutral element causing the cation to be smaller than its neutral atom.
Periodic Trends: Ionization & Electron Affinity
Ionization energy is defined as the energy required to add an electron to an atom or ion. Electron affinity is the energy change that occurs when an electron is added to an atom.
Solution
Ionization energy is defined as the energy required to remove an electron to an atom or ion.
Periodic Trends
The ionization energy and electron affinity of a group 2 element would be higher than that of a group 17 (7A) element.
Solution
Ionization energy is defined as the energy required to remove an electron from an atom.
Both ionization energy and electron affinity increase from:
- bottom-to-top, up a group.
- left-to-right, across a period.
Ionization energy generally increases from left-to-right across a period because it is harder (requires more energy) to remove an electron from elements that are close to having a full valence octet.
The ionization energy of a group 2 element would be lower than that of a group 17 (7A) element, in a similar period.
Both ionization energy and electron affinity increase from:
- bottom-to-top, up a group.
- left-to-right, across a period.
Ionization energy generally increases from left-to-right across a period because it is harder (requires more energy) to remove an electron from elements that are close to having a full valence octet.
The ionization energy of a group 2 element would be lower than that of a group 17 (7A) element, in a similar period.
Periodic Trends
Which element has the highest electronegativity?
Solution
Electronegativity is the ability of an atom to attract electrons when bonded.
Electronegativity increases left-to-right across a period, and bottom-to-top up a group.
Electronegativity increases left-to-right across a period, and bottom-to-top up a group.
An element with a low electron affinity will most likely also have
Background: Compounds
Which of the following would form a molecular compound?
Solution
Molecular compounds form by combining two nonmetals. Ionic compounds are formed by combining a metal + a nonmetal.
Background: Ionic Versus Covalent
An ionic bond forms with atoms that
Solution
Ionic bonds form when electrons are transferred from the positive cation to the negative anion.
Electron Dot Diagrams with Ionic Compounds
Draw the following molecules with electron dot diagrams:
Solution
Potassium Bromide, Calcium Dichloride, and Disodium Oxide
Potassium Bromide
Lewis Diagram: Potassium loses 1 electron as K+, bromine gains 1 electron as Br-.
Calcium Dichloride
Lewis Diagram: Calcium loses 2 electrons as Ca2+, each chlorine atom gains 1 electron as Cl-.
Disodium Oxide
Lewis Diagram: Each sodium loses 1 electron as Na+, oxygen gains 2 electrons as O2-.
Lewis Diagram: Potassium loses 1 electron as K+, bromine gains 1 electron as Br-.
Calcium Dichloride
Lewis Diagram: Calcium loses 2 electrons as Ca2+, each chlorine atom gains 1 electron as Cl-.
Disodium Oxide
Lewis Diagram: Each sodium loses 1 electron as Na+, oxygen gains 2 electrons as O2-.
Forming Covalent Compounds
Which of the following elements is most likely to form a covalent, molecular compound?
Solution
Covalent, molecular compounds form between two non-metals. Sodium and calcium are metals. Carbonate is a charged, polyatomic ion. Neon is a noble gas and doesn't bond... leaving Oxygen.
Identifying Covalent Compounds
Which of the following is a covalent compound?
Solution
Video
Covalent is two non-metals...
Lewis Structures: Intro
A Lewis structure for an element shows an arrangement of dots around the symbol of the element. These dots indicate the:
Lewis Structures of Covalent Compounds
Carbon would form the central atom in the molecule below.
Solution
CH2BrF


Lewis Structures of Covalent Compounds
Draw the Lewis Structures of the following molecules using the rules below.
| Step | What to Do |
| 1 | Arrange the atoms with the element that forms most bonds in the central position. |
| 2 | Add the total number of valence electrons to the central atom clockwise or counterclockwise. |
| 3 | Distribute the other atoms around the central atom and place one bonding pair of electrons between each atom. |
| 4 | Put remaining electrons as lone pairs on all atoms except the central atom, to a maximum octet of 8 electrons per atom. |
| 5 | If octet on central atom is incomplete, move peripheral lone pairs into bonding electrons that are shared with the central atom. |
| 6 | Make sure central and peripheral atoms have complete octets. Now if there are extra electrons, place these as lone pairs on the central atom (*exception to octet rule). |
CH4
Solution


H2O
Solution
Video


Br2
Solution


NH3
Solution
Video


N2H2
Solution


OH-
Solution


CO2
Solution
Video


Physical Properties of (Covalent) Molecular Compounds
Molecular compounds have a high melting point and are poor conductors of electricity when dissolved in water.
Solution
Molecular compounds have a low melting point and are poor conductors of electricity when dissolved in water.
Ionic compounds have a high melting and boiling point.
Ionic compounds have a high melting and boiling point.
Molecular Forces
Which of the following is not an intermolecular force?
Solution
Covalent is an intra-molecular bond...
The melting point and boiling point of ionic and covalent compounds can be explained with intramolecular forces.
Solution
The melting point of ionic and covalent compounds can be explained with intermolecular forces.
Intramolecular forces - within molecules - determine things like if a compound is likely to break apart and dissolve in water.
Intermolecular forces - between molecules - determine things like how much energy is required to melt or boil a substance because this requires energy to pull the different molecules away from adjacent molecules.
Intramolecular forces - within molecules - determine things like if a compound is likely to break apart and dissolve in water.
Intermolecular forces - between molecules - determine things like how much energy is required to melt or boil a substance because this requires energy to pull the different molecules away from adjacent molecules.
The van der Waals intermolecular force is composed of two forces: Dipole-dipole and London.
Solution
London forces are caused by the induced attraction of the electrons from one molecule to the positive centre in an adjacent molecule.
Dipole-dipole forces are caused by permanent dipoles of the molecules based on electronegativity differences. The van der Waals forces include: London and Dipole-dipole, but not Hydrogen bonding.
Dipole-dipole forces are caused by permanent dipoles of the molecules based on electronegativity differences. The van der Waals forces include: London and Dipole-dipole, but not Hydrogen bonding.
Which of the following molecules would only have London Dispersion Forces between the molecules.
Solution
Calculating the electronegativity difference between the atoms, you can see that N2 has the lowest (zero), so this molecule is non-polar covalent. Non-polar molecules do not have dipole-dipole forces, only London Dispersion.
Match the following intermolecular force properties...
Solution
Higher melting and boiling point
Lower melting and boiling point
Stronger force
Weaker force
Non-polar covalent
Polar covalent
Non-polar covalent has weaker intermolecular forces (London dispersion) vs polar covalent which has stronger intermolecular forces (London + Dipole + maybe even Hydrogen bonding...)
PERIODIC TRENDS
Electronegativity
Rank the elements in terms of increasing electronegativity (from bottom to top).
Solution
H, Br, C, F, P, Mg, K, I
Mg
Br
F
I
K
H
P
C
Electronegativity increases up and to-the-right on the periodic table.
Which of the following bonds has the highest electronegativity difference?
Solution
Video
Electronegativity difference is estimated by the distance across the periodic table, or calculated by subtracting the electronegativity of just one of each atom across a bond.
Br — Br = 0
C — O = 1.0
Ca — I = 1.66
Mg — O = 2.3
N — H = 0.9
Br — Br = 0
C — O = 1.0
Ca — I = 1.66
Mg — O = 2.3
N — H = 0.9
Neon has a higher electronegativity than Fluorine.
Solution
Rule notwithstanding, noble gases have extremely low electronegativity because of their full valence octet.
Electronegativity Difference (∆EN)
Complete the following table using the given electronegativities. Solution Video
| Element | Electronegativity |
| O | 3.44 |
| H | 2.2 |
| C | 2.55 |
| Al | 1.61 |
| Cl | 3.16 |
| Li | 0.98 |
| S | 2.58 |
| Na | 0.93 |
| F | 3.98 |
| Compound | Electronegativity Difference (∆EN) |
| H2O | 1.4 |
| CH4 | |
| AlCl3 | |
| Li2S | |
| NaF |
| Compound | Electronegativity Difference (∆EN) |
| H2O | 3.44 - 2.20 = 1.4 |
| CH4 | 2.55 - 2.20 = 0.35 |
| AlCl3 | 3.16 - 1.61 = 1.55 |
| Li2S | 2.58 - 0.98 = 1.6 |
| NaF | 3.98 - 0.93 = 3.05 |
Polarity Based on Electronegativity
Determine the polarity of the following molecules using the following guidelines. (* Indicates an exception to the guidelines).
| Electronegativity Difference Range: | 0 - 0.5 | 0.5 - 1.7 | 1.7 + |
| Bonding Result: | Non-polar Covalent | Polar Covalent | Ionic |
H2
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
Non-polar Covalent
CH4
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
Non-polar Covalent
LiF
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
Ionic
H2O
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
Polar Covalent
MgI2
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
*Ionic*
This molecule is ionic because it is made of a metal plus a non-metal, and it can dissolve into Mg2+ and 2I- in water as an electrolyte.
This molecule is ionic because it is made of a metal plus a non-metal, and it can dissolve into Mg2+ and 2I- in water as an electrolyte.
CO2
Solution
Video
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
*Non-polar Covalent*
This molecule is not polar covalent as the electronegativity would suggest (EN between 0.5 - 1.7). Instead it is non-polar because the molecule is symmetrical! Symmetry outranks the electronegativity difference. There is no net pull of the electrons shared in the covalent bonds in any direction...

This molecule is not polar covalent as the electronegativity would suggest (EN between 0.5 - 1.7). Instead it is non-polar because the molecule is symmetrical! Symmetry outranks the electronegativity difference. There is no net pull of the electrons shared in the covalent bonds in any direction...

Exceptions to Polarity Based on Electronegativity
Determine the polarity of the molecules that form between the metal Beryllium (Be) and the non-metals Hydrogen (H) and Carbon (C).
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
Covalent compounds typically occur between two non-metals... but Beryllium (Be) is an exception. Even though it is a metal, it can form covalent compounds with some non-metals...
Beryllium Hydride BeH2: Beryllium (Be) & Hydrogen (H)...
... is Polar Covalent
Beryllium Carbide BeC2: Beryllium (Be) & Carbon (C)...
...is Polar Covalent.
Beryllium Hydride BeH2: Beryllium (Be) & Hydrogen (H)... ... is Polar Covalent
Beryllium Carbide BeC2: Beryllium (Be) & Carbon (C)... ...is Polar Covalent.
Polarity
Hydrochloric acid (HCl) is a polar covalent molecule, rather than ionic.
Solution
The ∆EN between 0.5 to 1.7 indicates the molecule is polar covalent. The fact that this molecule physically dissolves into H+ and Cl- ions in water and conducts electricity does not make it ionic. As you see, it is possible for polar covalent molecules to dissociate into ions in solution.
Physical Properties, Boiling and Melting Point Based on Polarity
Using the polarity of the following molecules as a guide, order the molecules from low to high boiling points (from bottom to top).
Solution
CH4
H2O
CO2
LiF
Non-polar covalent < Polar covalent < Ionic.
Rank from low ∆EN to high ∆EN:
0.35 (CH4), 0.89 (CO2), 1.24 (H2O), 3.00 (LiF)
Rank from low ∆EN to high ∆EN:
0.35 (CH4), 0.89 (CO2), 1.24 (H2O), 3.00 (LiF)
Ionic Compounds
Which of the following ions would fit into this space indicated by 'Z' below?
Solution
MgZ2
Br-
Chemical Formulas
Complete the following table on paper, with the correct chemical formula for each compound.
Solution
Hint
Clear
Info
Ag+ Cu2+ Fe3+ Ti4+
Cl-
O2-
PO43- ( ) ( )
Incorrect Attempts:
CHECK
Balance the charges using the criss-cross method. Look up the charges for the polyatomic ions. Remember to reduce the subscripts by the greatest common factor (if any).
Ag+ Cu2+ Fe3+ Ti4+
Cl- AgCl CuCl2 FeCl3 TiCl4
O2- Ag2O CuO Fe2O3 TiO2
PO43- Ag3PO4 Cu3(PO4)2 FePO4 Ti3(PO4)4
Use the criss-cross method to balance the charges with subscripts on the ions. Subscripts with a common factor reduce. For example...
Cu2O2 --> CuO
Or...
Ti2O4 --> TiO2
| Ag+ | Cu2+ | Fe3+ | Ti4+ | |
| Cl- | ||||
| O2- | ||||
| PO43- | ( ) | ( ) |
| Ag+ | Cu2+ | Fe3+ | Ti4+ | |
| Cl- | AgCl | CuCl2 | FeCl3 | TiCl4 |
| O2- | Ag2O | CuO | Fe2O3 | TiO2 |
| PO43- | Ag3PO4 | Cu3(PO4)2 | FePO4 | Ti3(PO4)4 |
NAMING
When to Use All the Different Naming Systems
Compounds use different naming systems and it is important to know when to use each one.

Explain when the stock naming system is used, and how to use it. [3]
Solution
Video
Hint
Clear
Info
Incorrect Attempts:
CHECK
Look at the flow chart, given above
The STOCK system is used for multivalent (more than one possible charge like Cu1+/Cu2+, or Au1+/Au3+) metals in ionic compounds (ionic is a metal + a nonmetal). The second word does not use roman numerals, only the first word does. (Sometimes, rarely, the classical system is used instead of stock IUPAC naming)
(Side-note: the diatomic gases HOFBrINCl do not use any naming system, just name the element)
(Side-note: the diatomic gases HOFBrINCl do not use any naming system, just name the element)
Explain when the prefix naming system is used, and how to use it. [3]
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Look at the flow chart, given above
The PREFIX system is used for covalent compounds (covalent is two non-metals), not ionic compounds. A prefix is only used on the first word if 2 or more, and a prefix is always used on the last word. The second word ends with -ide. (Note that mono is only used in the second part of the name, never in the first part. CO2 is not monocarbon dioxide...)
Explain when the general naming system is used, and how to use it. [3]
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Look at the flow chart, given above
The GENERAL system is used if the ionic compound is not multivalent. Just name the first word like it appears on the periodic table and the second word ends with -ide (for example: Na20 is sodium oxide).
Naming Molecular/Covalent
Prefix Naming
Order the prefixes, with lowest at the top, by dragging them around.
Solution
deca
tri
penta
di
hepta
octa
tetra
hexa
nona
mono
1) Mono
2) di
3) tri
4) tetra
5) penta
6) hexa
7) hepta
8) octa
9) nona
10) deca
2) di
3) tri
4) tetra
5) penta
6) hexa
7) hepta
8) octa
9) nona
10) deca
Naming Covalent Molecules
Name the molecule using the prefix naming system: H2O
Solution
The prefix naming system is used in covalent compounds (between two non-metals). The prefix comes from the subscript number indicating number of each atom in the molecule.
_________ + _________
Tee first word uses a prefix if 2+ atoms...
The second word uses a prefix if 1+ atoms (always)...
The second word uses a prefix if 1+ atoms (always)...
Practice Naming Compounds: Molecular/Covalent
Name the following compounds using the prefix system.
CO
Solution
Video
Hint
Clear
Info
Incorrect Attempts:
CHECK
Determine the number of each element, and don't forget to add the suffix.
carbon monoxide
(not monocarbon monoxide - because mono never goes on the 1st word.)
(not monocarbon monoxide - because mono never goes on the 1st word.)
NO
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Determine the number of each element, and don't forget to add the suffix.
nitrogen monoxide
(Remember to only use mono on the second word, never the first).
(Remember to only use mono on the second word, never the first).
N2O
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Determine the number of each element, and don't forget to add the suffix.
dinitrogen monoxide
(drop the 'o' on the mono suffix)
(drop the 'o' on the mono suffix)
B2H6
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Determine the number of each element, and don't forget to add the suffix.
diboron hexahydride
(Still add the suffix -ide to the end of hydrogen = hydride)
(Still add the suffix -ide to the end of hydrogen = hydride)
CH4
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Determine the number of each element, and don't forget to add the suffix.
carbon tetrahydride
PCl5
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Determine the number of each element, and don't forget to add the suffix.
phosphorus pentachloride
CO2
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Determine the number of each element, and don't forget to add the suffix.
carbon dioxide
N2F4
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Determine the number of each element, and don't forget to add the suffix.
dinitrogen tetrafluoride
P2O5
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Determine the number of each element, and don't forget to add the suffix.
diphosphorus pentoxide
(drop the 'a' on the penta suffix)
(drop the 'a' on the penta suffix)
Practice Writing Formulas: Molecular/Covalent
Write the chemical formula for the following compounds.
Dihydrogen monosulfide
Solution
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
Incorrect Attempts:
CHECK
The prefix in-front of each element indicates the amount.
H2S
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
Nitrogen trihydride
Solution
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
Incorrect Attempts:
CHECK
The prefix in-front of each element indicates the amount.
NH3
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
Diphosphorus trisulfide
Solution
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
Incorrect Attempts:
CHECK
The prefix in-front of each element indicates the amount.
P2S3
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
Silicon tetrachloride
Solution
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
Incorrect Attempts:
CHECK
The prefix in-front of each element indicates the amount.
SiCl4
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
Nitrogen triiodide
Solution
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
Incorrect Attempts:
CHECK
The prefix in-front of each element indicates the amount.
NI3
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
Dinitrogen trioxide
Solution
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
Incorrect Attempts:
CHECK
The prefix in-front of each element indicates the amount.
N2O3
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
Sulfur trioxide
Solution
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
Incorrect Attempts:
CHECK
The prefix in-front of each element indicates the amount.
SO3
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
Trioxygen
Solution
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
Incorrect Attempts:
CHECK
The prefix in-front of each element indicates the amount.
O3
(common name: ozone)
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
(common name: ozone)
Correcting Common Mistakes
Find what's wrong with the following names or formulas, and correct them.
Mononitrogen monoxide
Solution
Video
Hint
Clear
Info
Incorrect Attempts:
CHECK
Where should prefixes be used?
Covalent compounds using the prefix naming system never have mono in the first name.
The correct name for NO:
nitrogen monoxide
(This is like carbon monoxide, which you may know is CO).
The correct name for NO: nitrogen monoxide (This is like carbon monoxide, which you may know is CO).
Dihydrogen sulfide
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Where should prefixes be used?
The last name always has a prefix, even if it's just one.
The correct name for H2S:
dihydrogen monosulfide
The correct name for H2S: dihydrogen monosulfide
C1F4
Solution
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
Incorrect Attempts:
CHECK
What subscripts should not be used?
Don't write subscripts of 1.
The correct formula is just:
CF4
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
The correct formula is just: CF4
Naming Ionic
Practice Naming Compounds: Ionic
Name the following ionic compounds. Remember that ionic compounds consist of a metal plus a non-metal.
MgO
Solution
Video
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
Mg2+O2-
Magnesium is not multivalent, so no roman numerals are required. Don't use prefix naming with ionic compounds (metal plus non-metal).
magnesium oxide
Magnesium is not multivalent, so no roman numerals are required. Don't use prefix naming with ionic compounds (metal plus non-metal). magnesium oxide
AlCl3
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
Al3+Cl1-
Aluminum is not multivalent, so no roman numerals are required.
aluminum chloride
Aluminum is not multivalent, so no roman numerals are required. aluminum chloride
InF3
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
indium fluoride
Na2O
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
sodium oxide
CaSe
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
calcium selenide
BaI2
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
barium iodide
ScF3
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
scandium fluoride
ZrBr4
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
zirconium bromide
Practice Writing Formulas: IONIC
Write the chemical formula for the following compounds.
Sodium sulfide
Solution
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
Incorrect Attempts:
CHECK
Start with the charges on the ions, and then use the criss-cross method to balance the charges
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
 |
Calcium sulfide
Solution
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
Incorrect Attempts:
CHECK
Start with the charges on the ions, and then use the criss-cross method to balance the charges
Using criss-cross with the ions... Ca2+ S2-
The 2s cancel, so there's one of each...
CaS
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
The 2s cancel, so there's one of each...
CaS
Silver fluoride
Solution
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
Incorrect Attempts:
CHECK
Start with the charges on the ions, and then use the criss-cross method to balance the charges
Using criss-cross with the ions... Ag1+ F1-
AgF
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
AgF
Zinc bromide
Solution
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
Incorrect Attempts:
CHECK
Start with the charges on the ions, and then use the criss-cross method to balance the charges
Using criss-cross with the ions... Zn2+ Br1-
ZnBr2
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
ZnBr2
Gallium oxide
Solution
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
Incorrect Attempts:
CHECK
Start with the charges on the ions, and then use the criss-cross method to balance the charges
Ga2O3
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
Lithium iodide
Solution
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
Incorrect Attempts:
CHECK
Start with the charges on the ions, and then use the criss-cross method to balance the charges
LiI
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
Germanium oxide
Solution
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
Incorrect Attempts:
CHECK
Start with the charges on the ions, and then use the criss-cross method to balance the charges
GeO2
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
Zirconium nitride
Solution
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
Incorrect Attempts:
CHECK
Start with the charges on the ions, and then use the criss-cross method to balance the charges. Use the most common ion charge.
Zr4+ + N3-...
Zr3N4
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
Zr3N4
Aluminum chloride
Solution
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
Incorrect Attempts:
CHECK
Start with the charges on the ions, and then use the criss-cross method to balance the charges. Use the most common ion charge.
AlCl3
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
Correcting Common Mistakes
Find what's wrong with the following names or formulas and correct them.
disodium monoxide
Solution
Video
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
Ionic compounds don't use the prefix naming system. The correct name would be:
sodium oxide
The roman numeral in, copper(II) sulphate, also known as copper(II) sulfate/sulphate, indicates that there are two atoms of copper per molecule.
Solution
The roman numeral indicates the charge (or oxidation number) of the multivalent cation.
scandium(III) phosphide
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
Don't use the stock roman numeral numbering system for monovalent elements - elements that only have one valence or possible charge.
Scandium can only ever have a 33+ charge and is implied in its name.
Correct name:
scandium phosphide
Correct name: scandium phosphide
Naming Polyatomics
Polyatomic Ions
How many atoms of Oxygen are in 2 molecules of aluminum nitrate?
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
The subscript, 3 multiplies to the 3 oxygens so 1 molecule of aluminum nitrate has 9 oxygens.
2 × Al(NO3)3
2 molecules of aluminum nitrate have 18 oxygens.
2 × Al(NO3)3 2 molecules of aluminum nitrate have 18 oxygens.
Polyatomic Anions
Which of the following polyatomic anions is sulphate (aka sulfate)?
Solution
Sulphate/Sulfate is SO42-
(Sulphite/Sulfite is SO32-)
The suffix (ending) with '-ate' has more oxygens because it ate more...
(Sulphite/Sulfite is SO32-)
The suffix (ending) with '-ate' has more oxygens because it ate more...
The oxides of chlorine and bromine form four different polyatomic anions with different numbers of oxygen atoms, x. Rank the naming system in the order of decreasing number of oxygens from top to bottom.
Solution
ClOx- BrOx-
_________-ite
per-_________-ate
hypo-_________-ite
_________-ate
In terms of the number of oxygens:
"Per-" is highest
"Hypo-" is lowest
"-ate" is higher
"-ite" is lower
ClO4–
perchlorate
ClO3–
chlorate
ClO2–
chlorite
ClO–
hypochlorite
BrO4–
perbromate
BrO3–
bromate
BrO2–
bromite
BrO–
hypobromite
"Per-" is highest
"Hypo-" is lowest
"-ate" is higher
"-ite" is lower
| ClO4– | perchlorate
|
| ClO3– | chlorate
|
| ClO2– | chlorite
|
| ClO– | hypochlorite
|
| BrO4– | perbromate
|
| BrO3– | bromate
|
| BrO2– | bromite
|
| BrO– | hypobromite
|
Making Ionic Compounds from Ions using the Criss-Cross Method (Including Polyatomic)
Compose the following ions into their salt/ionic compound by balancing the charges, and then name the compound. Put the formula of the compound in the box provided below, using proper capitalization and subscripts. [2]
Ca2+, PO43-
Solution
Video
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
Incorrect Attempts:
CHECK
Hint Unavailable
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
NH4+, CO32-
Solution
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
Incorrect Attempts:
CHECK
Hint Unavailable
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
Practice Naming Compounds: POLYATOMICS
Name the following compounds containing polyatomic ions.
Ca(NO3)2
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
calcium nitrate
(Note it's not calcium dinitrate because we don't use the prefix naming system on polyatomic ions).
(Note it's not calcium dinitrate because we don't use the prefix naming system on polyatomic ions).
Al2(SO3)3
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
aluminum sulphite (aka aluminum sulfite)
(Don't use prefix naming system: aluminum trisulfite, aluminum trisulphite)
(Don't use prefix naming system: aluminum trisulfite, aluminum trisulphite)
(NH4)SO4
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
ammonium sulphate (aka ammonium sulfate)
This is an ionic compound with polyatomic ions.
This is an ionic compound with polyatomic ions.
AgCN
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
silver cyanide
Zn(CH3COO)2, a.k.a. Zn(C2H3O2)2
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
zinc acetate
Al(HCO3)3
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
aluminum bicarbonate
Be3(PO3)2
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
beryllium phosphite
Zn(ClO2)
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
zinc chlorite
Mg(OH)2
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
magnesium hydroxide
KMnO4
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
potassium permanganate
Practice Writing Formulas: POLYATOMICS
Write the chemical formula for the following compounds with polyatomic ions.
Sodium bicarbonate
Solution
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
Incorrect Attempts:
CHECK
Hint Unavailable
Na+ + HCO3-
NaHCO3
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
NaHCO3
Magnesium perchlorate
Solution
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
Incorrect Attempts:
CHECK
Hint Unavailable
Mg2+ + ClO4-
Mg(ClO4)2
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
Mg(ClO4)2
Diammonium sulfide
Solution
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
Incorrect Attempts:
CHECK
Hint Unavailable
Diammonium is two (di) ammonium ions.
NH4+ + S2-
(NH4)2S
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
NH4+ + S2-
(NH4)2S
Tin(II) sulphite, aka Tin(II) sulfite
Solution
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
Incorrect Attempts:
CHECK
Hint Unavailable
Sn2+ + SO32-
SnSO3
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
SnSO3
Strontium cyanide
Solution
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
Incorrect Attempts:
CHECK
Hint Unavailable
Sr(CN)2
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
Silver phosphite
Solution
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
Incorrect Attempts:
CHECK
Hint Unavailable
Ag3PO3
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
Potassium hydroxide
Solution
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
Incorrect Attempts:
CHECK
Hint Unavailable
KOH
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
Aluminum acetate (C2H3O2)
Solution
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
Incorrect Attempts:
CHECK
Hint Unavailable
Al(C2H3O2)3
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
Correcting Common Mistakes
Find what's wrong with the following names or formulas and correct them.
nitrogen-tetrahydride bromide
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
Don't try to name polyatomic ions as molecular/covalent compounds. Use the name of the polyatomic ion. NH4Br is just ammonium bromide.
Sulphite (aka Sulfite): SO3
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
This may be a bit tricky because SO3 is not polyatomic when it's by itself. You can tell because it is not bound to a metal cation in-front, like __SO3.
SO3 is a covalent compound by itself - so you name it with the prefix system.
= sulfur trioxide
SO3 is a covalent compound by itself - so you name it with the prefix system.
= sulfur trioxide
Sodium Sulfur Oxide: Na2SO3
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
Na2SO3 is an ionic compound with a monovalent metal (and a polyatomic anion), so it is named using the general naming system.
= sodium sulphite (aka sodium sulfite).
= sodium sulphite (aka sodium sulfite).
Naming Multivalent
Naming Molecules: Ionic → Multivalent
Name the molecule using the stock naming system: Ni2O3
Solution
Video
Multivalent metals (metals that could have different charges like Ni2+ or Ni3+) need to have the charge represented by roman numerals without a space between the cation and roman numeral.
The oxygen anion oxygen begins with a prefix representing the number of atoms, and it ends with the suffix -ide indicating that it is an ion.
Stock: Nickel(III) Oxide
(Classical system: Nicklic Oxide)
The oxygen anion oxygen begins with a prefix representing the number of atoms, and it ends with the suffix -ide indicating that it is an ion.
Stock: Nickel(III) Oxide
(Classical system: Nicklic Oxide)
Practice Naming Compounds: Multivalent
Name the following compounds using the stock system.
CuS
Solution
Video
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
Cu2+S2-
Copper is multivalent (Cu1+, Cu2+) so roman numerals are required.
copper(II) sulfide
Copper is multivalent (Cu1+, Cu2+) so roman numerals are required.
copper(II) sulfide
CuCl2
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
copper(II) chloride
FeI3
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
Iron is multivalent (Fe2+, Fe3+) so use the stock naming system:
iron(III) iodide
iron(III) iodide
SnH4
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
Sn4+ + H-1
tin(IV) hydride
tin(IV) hydride
CoPO4
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
Co3+ + PO43-
cobalt(III) phosphate
cobalt(III) phosphate
AuP
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
Au3+ + P3-
gold(III) phosphide
gold(III) phosphide
TiS2
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
Ti4+ + S2-
titanium(IV) sulfide
titanium(IV) sulfide
Hg2O
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
Hg1- + O2-
mercury(I) oxide
mercury(I) oxide
Practice Writing Formulas: MULTIVALENT
Write the chemical formula for the following compounds with multivalent ions.
mercury(II) fluoride
Solution
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
Incorrect Attempts:
CHECK
Hint Unavailable
HgF2
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
copper(I) oxide
Solution
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
Incorrect Attempts:
CHECK
Hint Unavailable
Using criss-cross with the ions... Cu1+ O2-
Cu2O
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
Cu2O
copper(I) sulfide
Solution
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
Incorrect Attempts:
CHECK
Hint Unavailable
Using criss-cross with the ions... Cu1+ S2-
Cu2S
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
Cu2S
manganese(II) nitride
Solution
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
Incorrect Attempts:
CHECK
Hint Unavailable
Mn2+ + N3-
Mn3N2
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
Mn3N2
platinum(II) oxide
Solution
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
Incorrect Attempts:
CHECK
Hint Unavailable
Pt2+ + O2-
PtO
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
PtO
nickel(III) oxide
Solution
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
Incorrect Attempts:
CHECK
Hint Unavailable
Ni3+ + O2-
Ni2O3
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
Ni2O3
lead(IV) chloride
Solution
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
Incorrect Attempts:
CHECK
Hint Unavailable
Pb4+ + Cl-
PbCl4
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
PbCl4
antimony(V) phosphide
Solution
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
Incorrect Attempts:
CHECK
Hint Unavailable
Sb5+ + P3-
Sb3P5
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
Sb3P5
chromium(VI) oxide
Solution
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
Incorrect Attempts:
CHECK
Hint Unavailable
Cr6+ + O2-
Cr2O6
= CrO3
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
= CrO3
Correcting Common Mistakes
Find what's wrong with the following names or formulas and correct them.
iron(III) triiodide
Solution
Video
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
The stock naming system is used without using prefixes. The correct name:
iron(III) iodide
We know there must be 3 Iodide atoms for each Fe3+ because the charge on each iodide is only I1-.
Sn2O2
Solution
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
Incorrect Attempts:
CHECK
Hint Unavailable
Sn2+ + O2-
Using the criss-cross method, the subscripts will reduce to the lowest form:
= SnO
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
Using the criss-cross method, the subscripts will reduce to the lowest form:
= SnO
Naming Multivalent and Polyatomic
Practice Naming Compounds: Multivalent & Polyatomic
Name the following compounds using the systematic stock naming system.
Cu2+, SO42-
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
CuSO4
copper(II) sulphate, [aka copper(II) sulfate]
copper(II) sulphate, [aka copper(II) sulfate]
HgHCO3
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
Hg+1 + HCO3-
mercury(I) bicarbonate
mercury(I) bicarbonate
Fe3(PO3)2
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
Fe2+ + PO33-
iron(II) phosphite
iron(II) phosphite
Sn(CH3COO)4
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
Sn4+ + CH3COO1-
tin(IV) acetate
tin(IV) acetate
Au2SO4
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
Au+1 + SO42-
gold(I) sulphate, [aka gold(I) sulfate]
gold(I) sulphate, [aka gold(I) sulfate]
Co(ClO2)3
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
Co3+ + ClO2-
cobalt(III) chlorite
cobalt(III) chlorite
Pd(MnO4)4
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
Pd4+ + MnO4-1
palladium(IV) permanganate
palladium(IV) permanganate
Mn(NO3)2
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
Mn2+ + NO3-1
manganese(II) nitrate
manganese(II) nitrate
Practice Writing Formulas: Multivalent & Polyatomic
Write the chemical formula for the following compounds.
tin(II) bicarbonate
Solution
Tin (Sn) has a 2+ charge, bicarbonate (HCO3) has a 1- charge.
Use the criss-cross method to determine the subscripts on each ion.
Sn2+(HCO31-)
= Sn1(HCO3-)2
= Sn(HCO3-)2
Use the criss-cross method to determine the subscripts on each ion.
Sn2+(HCO31-)
= Sn1(HCO3-)2
= Sn(HCO3-)2
platinum(IV) nitrite
Solution
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
Incorrect Attempts:
CHECK
Balance the charges of the ions. Look up the polyatomic ions. Multivalent ions show their charge in parentheses.
Pt4+ + NO2-1
Pt(NO2)4
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
Pt(NO2)4
iron(III) cyanide
Solution
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
Incorrect Attempts:
CHECK
Balance the charges of the ions. Look up the polyatomic ions. Multivalent ions show their charge in parentheses.
Fe3+ + CN-
Fe(CN)3
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
Fe(CN)3
gold(I) acetate (C2H3O2-)
Solution
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
Incorrect Attempts:
CHECK
Balance the charges of the ions. Look up the polyatomic ions. Multivalent ions show their charge in parentheses.
Au+1 + C2H3O2- or CH3COO-
AuC2H3O2
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
AuC2H3O2
tungsten(V) hydroxide
Solution
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
Incorrect Attempts:
CHECK
Balance the charges of the ions. Look up the polyatomic ions. Multivalent ions show their charge in parentheses.
W5+ + OH-
W(OH)5
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
W(OH)5
vanadium(IV) bicarbonate
Solution
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
Incorrect Attempts:
CHECK
Balance the charges of the ions. Look up the polyatomic ions. Multivalent ions show their charge in parentheses.
V4+ + HCO3-1
V(HCO3)4
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
V(HCO3)4
manganese(VI) carbonate
Solution
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
Incorrect Attempts:
CHECK
Balance the charges of the ions. Look up the polyatomic ions. Multivalent ions show their charge in parentheses. Make sure to reduce the subscripts by the greatest common factor.
Mn6+ + CO32-
Mn2(CO3)6
Mn(CO3)3
(After criss-cross, reduce fully from 2:6 to 1:3)
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
Mn(CO3)3
(After criss-cross, reduce fully from 2:6 to 1:3)
tin(IV) phosphate
Solution
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
Incorrect Attempts:
CHECK
Balance the charges of the ions. Look up the polyatomic ions. Multivalent ions show their charge in parentheses.
Sn4+ + PO33-
Sn3(PO4)4
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
Sn3(PO4)4
Correcting Common Mistakes
Find what's wrong with the following names or formulas and correct them.
SnNO3 [with tin(IV)]
Solution
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
Incorrect Attempts:
CHECK
Hint Unavailable
Tin Sn4+, & nitrate, NO3-1...
Makes Sn(NO3)4
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
Makes Sn(NO3)4
Sodium(II) bromide
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
Sodium is not multivalent so it does not use the stock roman numeral naming system. It can only be Na+1.
The name is just sodium bromide.
The name is just sodium bromide.
Practice Naming Compounds: Multivalent & Polyatomic Oxyanions/Oxoanions, IUPAC
Modern, international IUPAC naming of compounds with polyatomic oxyanions, uses roman numerals with the oxyanion (as well as any multivalent cations). The only suffix used is 'ate'. Remember the roman numeral represents the oxidation number of the multivalent or non-oxide element. Name the following compounds, using full IUPAC notation.
E.g.) NO31-( # non-oxides )( oxidation state of non-oxide ) + ( # oxides )( oxidation state of oxide ) = polyatomic charge
∴ nitrate(V)
MgSO4
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
Mg2+ + SO42-
magnesium sulphate(VI), [aka magnesium sulfate(VI)]
magnesium sulphate(VI), [aka magnesium sulfate(VI)]
Na2SO2
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
Na1+ + SO22-
sodium sulphate(II), [aka sodium sulfate(II)]
sodium sulphate(II), [aka sodium sulfate(II)]
NH4ClO
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
NH4+ + ClO-
ammonium chlorate(I)
ammonium chlorate(I)
CuCrO4
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
Cu2+ + CrO42+
copper(II) chromate(VI)
copper(II) chromate(VI)
AlPO3
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
Al3+ + PO33-
aluminum phosphate(III)
aluminum phosphate(III)
Fe3(PO4)2
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
The subscript '2' on (PO43-)2 does not contribute to the oxidation number of the phosphorus.
Fe32+ + PO43-
iron(II) phosphate(V)
iron(II) phosphate(V)
(Ti)2(Cr2O7)3
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
'dichromate' is not used for the modern IUPAC naming of the polyatomic oxyanion.. use chromate(VI)
Ti3+ + Cr2O72-
titanium(III) chromate(VI)
titanium(III) chromate(VI)
Naming Hydrates
The name for the compound below is iron(II) sulphate hexahydrate. [note: sulphate is sometimes spelled sulfate].
Solution
FeSO4•6H2O
For hydrates, you have to combine the 'Stock' and the 'Prefix' naming systems.
Calculations with Hydrates, Intro
0.48 mol of hydrated barium chloride is heated to its anhydrous salt, BaCl2. If the mass of the hydrate is 117.2496 g, determine its molecular formula.
Solution
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
Incorrect Attempts:
CHECK
Hint Unavailable
MMBaCl2 = 208.23 g/mol
MMH2O = 18.02 g/mol
You can calculate the molar mass given the mass and amount of moles. Let 'n' be the molar coefficient of H2O in BaCl2·nH2O
Therefore there are 2 mols of H2O: BaCl2·2H2O
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
MMH2O = 18.02 g/mol
You can calculate the molar mass given the mass and amount of moles. Let 'n' be the molar coefficient of H2O in BaCl2·nH2O Therefore there are 2 mols of H2O: BaCl2·2H2O
Calculations with Hydrates
Epsom salts (magnesium sulphate heptahydrate, MgSO4·7H2O) are commonly used as bath salts. As an epsom salt is heated, to drive off the water, the mass continually decreases until the compound is anhydrous (lacking water). Under the right conditions, the salt, MgSO4 does not decompose.
The anhydrous compound contains less moles of MgSO4 than the fully hydrated compound.
Solution
The moles of MgSO4 are constant.
When the hydrate gives off water molecules, the amount of H2O changes while the MgSO4 stays constant because it cannot evaporate.
When the hydrate gives off water molecules, the amount of H2O changes while the MgSO4 stays constant because it cannot evaporate.
Find the amount of moles of water (H2O) in 100 g of epsom salt, given the molecular weights of MgSO4 is 120.3 g/mol, and 7H2O is 126 g/mol.
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
mol
Hint Unavailable
Find moles of the whole epsom salt compound, MgSO4·7H2O
Use molar ratios 7 H2O to 1 MgSO4·7H2O
[Note there is another method to get the answer using percent composition of water in the epsom, which is 51.157%, which is 51.157 g water, which is 2.84 mol water.]
Determine the mass of the anhydrous salt.
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
g
Hint Unavailable
Use molar ratios of 1 MgSO4 to 1 MgSO4·7H2O
Convert mol to mass:
[Note this could also be calculated using percent composition, not shown.]
ACIDS/BASES
Naming Acids: Rules
Match the example with the rule it falls under.
Solution
Acidic Halogens
HF, HCl, HBr, HI
Polyatomic anions ending in -ate
Nitrate NO3-,
Chlorate ClO3-,
Sulfate/Sulphate SO42-
Polyatomic anions ending in -ite
Nitrite NO2-,
Chlorite ClO2-,
Sulphite/Sulfite SO32-
Replace the suffix with the new suffix -ous, then add the word acid last.
Add the Hydro prefix to the front, replace part of the end with the suffix ic, then add the word acid last.
Replace the suffix with the new suffix -ic, then add the word acid last.
None of the above.
E.g.) Hydrofluoric acid, Hydrochloric acid, Hydrobromic acid, Hydroiodic acid
HF, HCl, HBr, HI
Nitrate NO3-, Chlorate ClO3-, Sulfate/Sulphate SO42-
Nitrite NO2-, Chlorite ClO2-, Sulphite/Sulfite SO32-
Naming Acids: State Matters
Determine the circumstance where the names below are used for HBr. [2]
Solution
Hydrogen bromide,
Hydrobromic acid
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
Use hydrogen bromide when the compound is a:
solid
Use hydrobromic acid when the compound is:
dissolved in water (aqueous)
Naming Acids: Polyatomic Anions
Name the following acids using the mnemonic given to help you remember the charges and number of oxygens in the main polyatomic anions ending in -ate. (The vowel number is the charge, and the consonant number is the number of oxygens).
NICK the CAMEL ate CLAMS for SUPPER in PHOENIX| Mnemonic | Consonants, Vowels | Formula | Name (-ate's) |
| Nick | 3, 1 | NO31- | Nitrate |
| Camel | 3, 2 | CO32- | Carbonate |
| Clam | 3, 1 | ClO31- | Chlorate |
| Supper | 4, 2 | SO42- | Sulfate/Sulphate |
| Pheonix | 4, 3 | PO43- | Phosphate |
H2SO4
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
Ions ending in -ate take the suffix -ic
sulfuric acid
sulfuric acid
H2SO3
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
Ions ending in -ite take the suffix -ous
sulfurous acid
sulfurous acid
HNO3
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
Ions ending in -ate take the suffix -ic
nitric acid
nitric acid
HNO2
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
Ions ending in -ite take the suffix -ous
nitrous acid
nitrous acid
HClO3
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
Ions ending in -ate take the suffix -ic
chloric acid
(Perchloric = ClO4)
chloric acid
(Perchloric = ClO4)
HClO2
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
Ions ending in -ite take the suffix -ous
chlorous acid
chlorous acid
H3PO4
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
Ions ending in -ate take the suffix -ic
phosphoric acid
phosphoric acid
H3PO3
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
Ions ending in -ite take the suffix -ous
phosphorous acid
phosphorous acid
H2CO3
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
Ions ending in -ate take the suffix -ic
carbonic acid
carbonic acid
H2CO2
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
Ions ending in -ite take the suffix -ous
carbonous acid
(Aside: common names are formic acid or methanoic acid)
carbonous acid
(Aside: common names are formic acid or methanoic acid)
Naming Bases
You can easily recognize bases that have hydroxide (OH-) ions in them. Name the following bases. Remember the proper syntax of naming: lowercase and proper spacing --- for example, Ti(OH)4 = titanium(IV) hydroxide.
KOH
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
potassium hydroxide
NH4OH
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
ammonium hydroxide
Mg(OH)2
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
magnesium hydroxide
(Do not use prefix naming, like di, in bases)
(Do not use prefix naming, like di, in bases)
Fe(OH)3
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
Multivalent ions, like Fe2+, Fe3+ use the stock naming system.
iron(III) hydroxide
(Don't use prefix naming — like mono, di, tri — in bases)
iron(III) hydroxide
(Don't use prefix naming — like mono, di, tri — in bases)
Pb(OH)2
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
Multivalent ions, like Pb2+, Pb4+ use the stock naming system.
lead(II) hydroxide
(Don't use prefix naming — like mono, di, tri — in bases)
lead(II) hydroxide
(Don't use prefix naming — like mono, di, tri — in bases)
Recognizing Bases in Grade 11
Which of the following is not a base?
Solution
Bases have different number of hydroxide (OH-) ions in the formula.
The exception is ammonia, NH3, which is the base form of ammonium, NH4+.
The exception is ammonia, NH3, which is the base form of ammonium, NH4+.
NUCLEAR CHEM
Radioactive Decay
Uranium (U) decays into a Thorium (Th) isotope and what other kind of particle?
Solution
is an alpha particle
Nuclear Reaction
The following reaction is nuclear fusion.
Solution
The reaction is nuclear fission. Fission is the splitting of a large nucleus into smaller nuclei.
Flashcards: Matter and Bonding
| How many neutrons in |
| 31 |
| What is an energy level? |
| An orbit or shell corresponding to the period number, on which electrons are located. |
| What is the trend for increasing electronegativity on the periodic table? |
| Up a group, and right across a period |
| What is the trend for increasing electron affinity on the periodic table? |
| Up a group, and right across a period |
| What is the trend for increasing atomic radius on the periodic table? |
| Down a group, and left across a period |
| What is the trend for increasing ionization energy on the periodic table? |
| Up a group, and right across a period |
| How does the atomic radius of elemental fluorine (F) compare to its ion form (F-)? |
| The fluorine ion has a larger radius because the extra electron causes more repulsion in the electron cloud and expands the size of the radius |
| How does the atomic radius of a cation compare to its neutral elemental form? |
| The atomic radius of the cation is always smaller than the neutral atom. |
| How are ionization energy, electronegativity, and electron affinity related? |
| Electronegativity is the attraction of the electrons by the element, which is the same trend for electron affinity. Ionization energy is the energy required to remove an electron, which is high for elements with high electronegativity and electron affinity |
| What is the difference between an ionic compound and a molecular compound? |
| Ionic compounds are a metal + a non-metal. Molecular compounds are two non-metals. (another name for molecular compound is covalent compound) |
| What is the difference between an ionic compound and an alloy? |
| An alloy is composed of two or more metals while an ionic compound is composed of a metal + a non-metal. Alloys do not form intramolecular bonds like in ionic bonds. |
| What type of compounds are: SO2 or NF3? |
| Covalent/Molecular |
| What type of compound is Sodium Fluoride? |
| Ionic compound |
| What does a lewis dot diagram indicate? |
| The arrangement of bonding and non-bonding (lone pair) electrons in an atom or compound. |
| When drawing lewis structures, what is the next step to do if the octet on a central atom is incomplete? |
| Move peripheral lone pairs on adjacent atoms into bonding electrons that are shared with the central atom. |
| What are some of the main physical properties of molecular compounds? |
| Molecular compounds have a low melting point and are poor conductors of electricity when dissolved in water. |
| What are the three (four) intermolecular forces you have seen so far? |
| London, Dipole-dipole, van der Waals (London + Dipole-Dipole), and Hydrogen bonding |
| A molecule with a 0.6 electronegativity difference would be what type of polarity? |
| Polar covalent. |
| Name the molecule with the stock naming system: Fe2O3 |
| Iron (III) Oxide |
| Name the molecule with the prefix naming system: Fe2O3 |
| Diiron trioxide |
Quantities in Chemical Reactions
Quantities in Molecules
Which is the coefficient in the following molecule?
Solution
2 Al(NO3)3
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
The coefficient is the number in front... 2
Balancing Chemical Equations
Which of the following equations is balanced?
Solution
Conventionally, a balanced equation contains whole number coefficients.
The equation below is not in whole number ratios, and while it is technically balanced, it is not considered standard form:
N2(g) + 2.5O2(g) → N2O5(g)
The equation below is not in whole number ratios, and while it is technically balanced, it is not considered standard form:
N2(g) + 2.5O2(g) → N2O5(g)
Balancing Chemical Equations
Balance the following chemical reaction equation.
Solution
Hint
Clear
Info
C6H14 + O2 CO2 + H2O
Incorrect Attempts:
CHECK
Hint Unavailable
2C6H14 + 19O2 12CO2 + 14H2O
Balancing Chemical Equations
Balance the following equations. Balance the metal, or carbon, or first non-metal first. Then balance the hydrogens (if any). Last, balance the diatomic molecule like O2.
N2 + O2 N2O5
Solution
Hint
Clear
Info
N2 + O2 N2O5
Incorrect Attempts:
CHECK
Generally: balance metals first (if any), balance polyatomic ions as whole units, and balance diatomic molecules last.
C6H12O6 + O2 CO2 + H2O
Solution
Video
Hint
Clear
Info
C6H12O6 + O2 CO2 + H2O
Incorrect Attempts:
CHECK
Generally: balance metals first (if any), balance polyatomic ions as whole units, and balance diatomic molecules last.
C6H14 + O2 CO2 + H2O
Solution
Hint
Clear
Info
C6H14 + O2 CO2 + H2O
Incorrect Attempts:
CHECK
Generally: balance metals first (if any), balance polyatomic ions as whole units, and balance diatomic molecules last.
Al(OH)3 + H2CO3 Al2(CO3)3 + H2O
Solution
Video
Hint
Clear
Info
Al(OH)3 + H2CO3 Al2(CO3)3 + H2O
Incorrect Attempts:
CHECK
Generally: balance metals first (if any), balance polyatomic ions as whole units, and balance diatomic molecules last.
Word Equations
Write balanced chemical equations for each of the following word equations and make sure to include the physical states.
Heating solid iron in the presence of oxygen.
Solution
Solid bromine reacts with sodium iodide.
Solution
Bromine is higher on the activity series than iodine, therefore it can displace the iodine.
Solid magnesium sulfate heptahydrate is heated for a long period of time.
Solution
Single Displacement Reactions: A + BC → B + AC
Complete the following single displacement reactions.
Halogens Replace Halogens:
Solution
F2(g) + NaCl(aq) gas + salt
Hint
Clear
Info
+ →+
Incorrect Attempts:
CHECK
Write the compounds first, then balance. Make sure to include physical states: (s), (l), (g), (aq).
Metal Replaces Hydrogen, in Metal + Acid:
Solution
Mg(s) + HCl(aq) gas + salt
Hint
Clear
Info
+ →+
Incorrect Attempts:
CHECK
Write the compounds first, then balance. Make sure to include physical states: (s), (l), (g), (aq).
Metal Replaces other Metal, in Metal + Ionic Compound:
Solution
Video
Al(s) + Cu(NO3)2(aq) gas + salt
Hint
Clear
Info
+ ( ) →+ ( )
Incorrect Attempts:
CHECK
Write the compounds first, then balance. Make sure to include physical states: (s), (l), (g), (aq).
Double Displacement Reactions: AB + CD → AD + CB
The reaction between sodium carbonate and copper (II) sulfate/sulphate proceeds as follows. Determine the identity of compound 'Z' below.
Double Displacement Reactions: AB + CD → AD + CB
Complete the following double displacement reaction. Remember to use what you know about the solubility guidelines.
NaClO3 (aq) + MgF(aq) ______________
Solution
Hint
Clear
Info
+ →+ ( )
Incorrect Attempts:
CHECK
Write the compounds first, then balance. Make sure to include physical states: (s), (l), (g), (aq).
2NaClO3(aq) + MgF2(aq) 2NaF(aq) + Mg(ClO3)2(aq)
Metals Replace Metals
Solution
AgNO3(aq) + KI(aq) aqueous compound + solid compound
Hint
Clear
Info
+ →+
Incorrect Attempts:
CHECK
Write the compounds first, then balance. Make sure to include physical states: (s), (l), (g), (aq).
Reactivity
Use the reactivity series table for the questions below.
| Reactivity Ranking | Element |
| 1 (Most Reactive) | Lithium, Li |
| 2 | Potassium, K |
| 3 | Barium, Ba |
| 4 | Calcium, Ca |
| 5 | Sodium, Na |
| 6 | Magnesium, Mg |
| 7 | Aluminum, Al |
| 8 | Zinc, Zn |
| 9 | Chromium, Cr |
| 10 | Iron, Fe |
| 11 | Cadmium, Cd |
| 12 | Cobalt, Co |
| 13 | Nickel, Ni |
| 14 | Tin, Sn |
| 15 | Lead, Pb |
| 16 | Hydrogen, H |
| 17 | Copper, Cu |
| 18 | Mercury, Hg |
| 19 | Silver, Ag |
| 20 | Platinum, Pt |
| 21 (Least Reactive) | Gold, Au |
Which of the following could displace the Chromium in Cr(III) Chloride.
Solution
Calcium ranks higher on the reactivity (activity) series and is more reactive than Chromium. Calcium will displace Chromium in a single displacement reaction.
Predict which reaction (below) will occur spontaneously (by itself).
Solution
Calcium is higher on the reactivity series so it will displace the less reactive Iron.
The reaction proceeds: Ca(s) + FeBr3 (aq) → Fe(s) + CaBr2 (aq).
Don't forget to balance: 3Ca(s) + 2FeBr3 (aq) → 2Fe(s) + 3CaBr2 (aq)
The reaction proceeds: Ca(s) + FeBr3 (aq) → Fe(s) + CaBr2 (aq).
Don't forget to balance: 3Ca(s) + 2FeBr3 (aq) → 2Fe(s) + 3CaBr2 (aq)
Reactivity
Using your understanding of the reactivity (activity) series, explain the following concepts below.
Pure Silver and Gold never tarnishes.
Solution
Silver and Gold are two of the most unreactive metals and they are at the bottom of the activity series.
Zinc is used to coat iron materials to protect from rusting.
Solution
Zinc is more reactive than iron and reacts first with the oxygen in air to produce a protective zinc oxide coating around the iron. This coating prevents iron from being exposed in air to rust. The process of coating iron with zinc is common and is called galvanization.
Chemical Equations Based on Activity Series
For each of the scenarios below, predict the products of the chemical reactions, balance, and classify the type of reaction.
Iron powder is mixed into nitric acid.
Solution
Iron is more reactive than Hydrogen and will displace it from the nitric acid.
Fe(s) + 2HNO3 (aq) → H2 (g) + Fe(NO3)2 (aq)
Single displacement.
When potassium is dropped into water, a gas along with potassium hydroxide is formed.
Solution
Potassium is more reactive than Hydrogen and will displace it from the water.
2K(s) + 2H2O(l) → H2 (g) + 2KOH(aq)
Single displacement.
Ethanol (C2H6O(l)) is burned in excess oxygen.
Solution
Combustion reactions in excess oxygen always form water and carbon dioxide. Remember that 'excess oxygen' means lots of oxygen on the reactant side of your equation...
C2H6O(l) + 3O2 (g) → 3H2O(g) + 2CO2 (g)
Combustion reaction.
Molecular Weight (Molar Mass)
Determine the molar mass of...
CH4
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
g/mol
Hint Unavailable
1 Carbon + 4 Hydrogens
AgNO3
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
g/mol
Hint Unavailable
1 Ag + 1 N + 3 O's
Ca(NO3)2.
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
g/mol
The subscript on parentheses ()2 applies to all elements within the parentheses.
Isotope Abundances: Abundance
The average atomic mass of an element depends on the number of protons and neutrons only.
Solution
The atomic mass of an element depends on the amount of protons and neutrons and also depends on the relative abundance of the different isotopes of the element, found in nature.
If the relative abundance (%) of an heavy isotope is high or low, this will affect the magnitude of the atomic mass.
If the relative abundance (%) of an heavy isotope is high or low, this will affect the magnitude of the atomic mass.
Titanium has five different isotopes in various proportions: 8.25% 46Ti, 7.44% 47Ti, 73.72% 48Ti, 5.41% 49Ti, and 5.18% 50Ti. The average atomic mass of Titanium will be closest to
Solution
Because 48Ti is most abundant at 73.72%
Isotope Abundances: Average Atomic Mass
For our consideration, if silicon is simplified to have three different isotopes, then determine the average atomic mass. Show your work. Solution
| Isotope | Relative Abundance |
| 28Si | 92.2 % |
| 29Si | 4.7 % |
| 30Si | 3.1 % |
Isotope Abundances
Element X has two isotopes, 23X and 21X. If the average atomic mass of element X is 21.5 g/mol, find the percent abundance of each isotope. [2]
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
Let 'x' be the percent abundance of 23X.
Let (100 - x) be the percent abundance of 21X.
Let (100 - x) be the percent abundance of 21X.
Avogadro and The Mole
Which of the following statements is false regarding Avogadro's number (6.02 × 1023)?
Solution
Avogadro's number stands for 6.02 × 1023 .
These "things" depend on the context and can be atoms, particles, molecules, compounds, etc...
These "things" depend on the context and can be atoms, particles, molecules, compounds, etc...
Molar Ratios
Determine the missing quantity in the following table. Solution
| 2 NH3 | 1 CH4 | |
| Mole Ratio | 2 | 1 |
| Molecules | 2 | 1 |
| Molecules in 1 mole | 6.02 × 1023 |
| 2 NH3 | 1 CH4 | |
| Mole Ratio | 2 | 1 |
| Molecules | 2 | 1 |
| Molecules in 1 mole | 1.2 × 1024 | 6.02 × 1023 |
The 3 M's: Mass, Moles, and Molecules
Carbon dioxide (CO2) has a molar mass of 44g/mol.
Determine the number of moles in 88.0g of carbon dioxide.
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
mol
Hint Unavailable
Determine the number of molecules in the sample above.
Solution
Hint
Clear
Info
× 10
Incorrect Attempts:
CHECK
molecules
Hint Unavailable
The 3 M's: Mass, Moles, and Molecules
Magnesium difluoride (MgF2) has a molar mass of 62.3 g/mol.
Determine the number of moles in 1.85 × 1022 molecules of MgF2
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
mol
Hint Unavailable
Determine the mass in the sample above.
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
g
Hint Unavailable
Avogadro and The Mole
Given the atomic mass of carbon is 12.01 grams per mole, determine the mass of 6.02 × 1023 carbon atoms.
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
g
Hint Unavailable
6.02 × 1023 is one mole of carbon atoms.
See ...
Determine the number of mols of carbon in 3.01 × 1023 molecules of nickel(II) bicarbonate, Ni(HCO3)2.
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
mol
Hint Unavailable
Use the stoichiometric ratio to convert moles of Ni(HCO3)2 to moles of 'C'. 1 molecule contains 2 carbon atoms.
Percent Composition
A 10.5 mol sample of H2CO3 has the same percent composition of hydrogen as a 3.0 mol sample. Show your work.
Percent Composition
Determine percentage composition, by mass of each element in Fe(NO3)2.
Solution
Hint
Clear
Info
% Fe = %
% N = %
% O = %
Incorrect Attempts:
CHECK
Hint Unavailable
% N = %
% O = %
Empirical and Molecular Formulas
Both C6H12 and C2H4 have the same empirical formula.
Solution
Because when the molecular formulas are both reduced, they will have the same ratio of C to H, which is the same empirical formula.
Which of the following is a molecular, but not an empirical formula?
Solution
An empirical formula is the most simplified (reduced) ratio or proportion of the atoms in a molecule.
Empirical and Molecular Formulas
Given the empirical formula and molecular weight of the compound, it is possible to determine the molecular formula.
Solution
True
If the molecular weight of a compound is 586.64 g/mol, determine the molecular formula given the empirical formula, C2H3NO2.
Solution
Hint
Clear
Info
C H N O
Incorrect Attempts:
CHECK
Hint Unavailable
The molecular formula is a multiple, 'x' of the empirical formula,
C2(x)H3(x)N1(x)O2(x)
Calculate the factor x, using the MM of the empirical formula (see periodic table), 73.06 g/mol,
The molecular formula is a factor of 8 greater than the empirical formula:
C16H24N8O16
Empirical and Molecular Formulas
One mole of a molecular compound contains 7.224 × 1024 atoms of hydrogen. Determine the molecular formula, given the empirical formula, CH2S.
Solution
Hint
Clear
Info
C H S
Incorrect Attempts:
CHECK
Hint Unavailable
This requires some careful understanding and consideration of what the units represent.
This shows that there are 12 atoms of 'H' in a molecule of the molecular formula, so you should be able to see that the empirical formula is increased by a factor of 6, (H2 → × 6 → H12)
C6H12S6
Empirical and Molecular Formulas with Percent Composition
Analysis of an organic molecule shows that it contains 61.02% carbon, 11.86% hydrogen and 27.12% oxygen.
Determine the empirical formula of the molecule.
Solution
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
Incorrect Attempts:
CHECK
Hint Unavailable
Assume 100 g, and convert to moles,
= C3H7O
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
Given the molar mass of the compound is 118.1 g/mol, determine the molecular formula.
Solution
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
Incorrect Attempts:
CHECK
Hint Unavailable
Determine the molar mass of C3H7O
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
Empirical Formulas with Magnesium in Lab
You want to experimentally determine the empirical formula of magnesium oxide. In the lab at your school, you weigh 3.0 g of solid magnesium and burn it completely in a crucible. You know this forms magnesium oxide, and you determine the mass is 4.9 g. Assuming no other side or intermediate reactions, determine the empirical formula.
Solution
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
Incorrect Attempts:
CHECK
Hint Unavailable
The mass of magnesium is conserved, and only oxygen has been added. The mass of oxygen that was added is,
Next, convert mass to moles, and divide by the lowest,
Mass Mols Ratio Subscript
1.9 g Oxygen = 1
3.0 g Magnesium ≈ 1
Therefore the empirical formula is MgO.
⁰
¹
²
³
⁴
⁵
⁶
⁷
⁸
⁹
⁻
⁺
⁽
⁾
₀
₁
₂
₃
₄
₅
₆
₇
₈
₉
₋
₊
₍
₎
| Mass | Mols | Ratio | Subscript |
| 1.9 g Oxygen | = 1 | ||
| 3.0 g Magnesium | ≈ 1 |
Empirical Formulas with Hydrates
A 25.0 g sample of hydrated sodium sulfate/sulphate is heated to drive off the water, without decomposing the ionic compound. This leaves 15.3 g of the anhydrous ionic compound. Determine the coefficient of water in the empirical formula of the hydrate, Na2SO4·__H2O
Solution
coefficient =
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
The mass of the ionic compound, sodium sulfate/sulphate (Na2SO4), and water are both conserved. We can calculate the mass of the water that was lost from the starting hydrated compound.
Next, convert mass to moles, and divide by the lowest,
Mass Mols Ratio Subscript
9.7 g Water ≈ 5
15.3 g Sodium sulfate/sulphate = 1
Therefore the empirical formula is Na2SO4·5H2O
| Mass | Mols | Ratio | Subscript |
| 9.7 g Water | ≈ 5 | ||
| 15.3 g Sodium sulfate/sulphate | = 1 |
Limiting Reagent, Intro
Given 50 slices of bread, 24 leaves of lettuce, and 84 slices of cheese, determine the limiting ingredient in the following recipe:
Solution
2 Bread + 1 Lettuce + 3 Cheese → 1 Sandwich
Bread
Can make 50/2 = 25 sandwiches with the starting amount.
Lettuce
Can make 24/1 = 24 sandwiches with the starting amount.
Cheese
Can make 84/3 = 28 sandwiches with the starting amount.
Using this algorithm, the limiting reagent is the lowest value. Therefore the lettuce is the limiting ingredient.
Can make 50/2 = 25 sandwiches with the starting amount.
Lettuce
Can make 24/1 = 24 sandwiches with the starting amount.
Cheese
Can make 84/3 = 28 sandwiches with the starting amount.
Using this algorithm, the limiting reagent is the lowest value. Therefore the lettuce is the limiting ingredient.
In the imaginary reaction below, 10 mol of AB, 15 mol of CD, 6 mol of EF, and 16.5 mol of G react, which reactant is limiting?
Solution
4AB + 5CD + EF + 3G → 2HI + J + 8K
The lowest numer is limiting.
AB: 10/4 = 2.5
CD: 15/5 = 3.0
EF: 6/1 = 6
G: 16.5/3 = 5.5
Therefore AB is the limiting reactant
AB: 10/4 = 2.5
CD: 15/5 = 3.0
EF: 6/1 = 6
G: 16.5/3 = 5.5
Therefore AB is the limiting reactant
Limiting Reactant
Which is the limiting reactant in the combustion reaction of ethanol, C2H6O, below given 0.20 mol of ethanol and 0.30 mol of oxygen.
Solution
Molar Ratios Without Limiting Reactant
100g of octane fuel, C8H18, burns in excess oxygen to create energy, water, and carbon dioxide.
Find the amount of moles of octane.
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
mol
Hint Unavailable
Determine the balanced equation,
Molar mass of octane is 114.26g/mol.
Molar mass of octane is 114.26g/mol.
Write the balanced chemical equation to determine the theoretical yield of water, in moles, using the molar ratio of octane to water.
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
mol
Hint Unavailable
Based on the balanced equation,
The stoichiometric ratio is,
What mass of water is produced?
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
g
Hint Unavailable
Molar mass of water is 18.01g/mol.
Stoichiometry: Molar Ratios With Limiting Reactant
Starting with 18mol of A and 8mol of B, how many moles of excess reactant is left over when the reaction is complete?
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
mol
Hint Unavailable
The limiting is the lowest value from the ratio of the mols available, to the mols required by the coefficient in the equation:
Therefore 'B' is limiting..
Mols of 'A' used:
Mols of 'A' remaining
Mols of 'A' used: Mols of 'A' remaining
Stoichiometry: Molar Ratios With Limiting Reactant
53.06 g of solid iron is dipped into a beaker containing 113.36 g nitric acid, bubbles are observed from the single displacement reaction.
Write the balanced chemical equation to find the number of moles of each reactant.
Solution
Hint
Clear
Info
mol Fe =
mol HNO3 =
Incorrect Attempts:
CHECK
Hint Unavailable
mol HNO3 =
Determine the limiting reactant assuming all reactants can react fully.
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
The limiting reactant is the lowest value of the ratio of moles to the coefficient from the equation.
The ratio for Nitric Acid is lower, therefore Nitric Acid is the limiting reactant.
The ratio for Nitric Acid is lower, therefore Nitric Acid is the limiting reactant.
Calculate the mass of the gas produced.
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
g
Hint Unavailable
Always calculate the amount of a product with the limiting reactant, which in this case is nitric acid.
Stoichiometry: Molar Ratios With Limiting Reactant
Determine the mass of barium required to completely displace copper in 20.0 g of copper(II) sulfate/sulphate.
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
g
Hint Unavailable
Stoichiometry: Molar Ratios With Limiting Reactant
A 16.19 g sample of solid aluminum reacts with 254.76 g of silver nitrate.
Determine the limiting reagent.
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
Determine the mass of excess reactant left over when the reaction is complete.
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
g
Hint Unavailable
All silver nitrate used. Find moles of aluminum remaining:
Stoichiometry: Molar Ratios With Hydrate
Thermonitrite, a soluble hydrate of sodium carbonate, reacts with hydrochloric acid to produce a salt, carbon dioxide gas, and water.
If 14.531 g of carbon dioxide is produced from 40.96 g of thermonitrite, determine the chemical formula of thermonitrite, hence the value of X.
Solution
X =
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
Write the chemical formula and balance. To balance, see that for 'X' amount of hydrated water, there are (2 + X) waters produced. This is a tricky part of the question.
Determine the amount of mol of CO2 (g)
Use the stoichometry ratio to find: amount of moles of thermonitrite.
Use algebra to solve for 'x', with the equation below:
Therefore the chemical formula is Na2CO3·1H2O, since X = 1.
Stoichiometry: Molar Ratios With Hydrate
The hydrate, BaCl2 · XH2O contains 56.22% Barium by mass. Determine the unknown coefficient, 'X' of the hydrate.
Solution
X =
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
! Assume 100 grams. So 56.22% Barium equals 56.22 g Barium...
Determine the amount of moles of Barium,
Since there is 1 Ba : 2 Cl, then there are 0.818758 mol of Cl, from 2(0.409379).
Calculate the remaining mass of water, since you assumed the total is 100 grams.
Determine the amount of moles of H2O
The calculations above are stored into the table below:
Component Mass (g) Moles (mol) Amount (#)
Ba 56.22 0.409379 0.409379/0.409379 = 1
Cl 29.02496 2(0.409379) 2(0.409379)/0.409379 ≈ 2
H2O 14.755 0.8188 0.8188/0.409379 ≈ 2
Finally divide the amount of moles by the lowest value, 0.409379 and this gives the amount of each component in the hydrate. Therefore the compound is BaCl2 · 2H2O.
An alternate way of solving this is to use molar masses, and to let 'x' be the molar mass of water:
There are 2 mols of H2O. Therefore the compound is BaCl2 · 2H2O.
Determine the amount of moles of Barium, Since there is 1 Ba : 2 Cl, then there are 0.818758 mol of Cl, from 2(0.409379). Calculate the remaining mass of water, since you assumed the total is 100 grams. Determine the amount of moles of H2O The calculations above are stored into the table below:
| Component | Mass (g) | Moles (mol) | Amount (#) |
| Ba | 56.22 | 0.409379 | 0.409379/0.409379 = 1 |
| Cl | 29.02496 | 2(0.409379) | 2(0.409379)/0.409379 ≈ 2 |
| H2O | 14.755 | 0.8188 | 0.8188/0.409379 ≈ 2 |
An alternate way of solving this is to use molar masses, and to let 'x' be the molar mass of water: There are 2 mols of H2O. Therefore the compound is BaCl2 · 2H2O.
Theoretical and Actual Yield
The actual yield is what we calculate, and the theoretical yield is what is measured in the lab.
Solution
The actual yield is determined experimentally in the lab, and the theoretical yield is calculated. The actual yield is typically lower than the theoretical yield.
Percent Yield: Theoretical and Actual Yield
If 56.0g of nitrogen is reacted with excess hydrogen and measured to yield 66.93g of ammonia, determine the percent yield of the synthesis reaction.
Solution
Use mass and not moles.
Percent Yield: Theoretical and Actual Yield
If the percent yield of the sugar in cellular respiration is 95% and the actual yield of the sugar is 100.0g, determine the mass of carbon dioxide that reacted.
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
g
Hint Unavailable
Percent Yield
It is possible to have percent yields greater than 100%.
Solution
Yes it is possible. This situation happens sometimes when:
- There is some solvent left over in the product, or if foreign objects got into the product.
- Impure reactants can leave traces of remnants or byproducts in the product.
- Miscalculations of readings of volume, mass, etc...
- ...
- There is some solvent left over in the product, or if foreign objects got into the product.
- Impure reactants can leave traces of remnants or byproducts in the product.
- Miscalculations of readings of volume, mass, etc...
- ...
Explain why the percent yield is typically less than 100%.
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
- Reactions usually reach equilibrium and 'stop' before everything is reacted.
- Transferring the reactants between different containers can leave some behind.
- Miscalculations of readings of volume, mass, etc...
- Sometimes it takes considerable time for a reaction to proceed, and the reaction in a lab is often cut short on time.
- ...
- Reactions usually reach equilibrium and 'stop' before everything is reacted.
- Transferring the reactants between different containers can leave some behind.
- Miscalculations of readings of volume, mass, etc...
- Sometimes it takes considerable time for a reaction to proceed, and the reaction in a lab is often cut short on time.
- ...
Flashcards: Quantitative Chemistry
| How many moles of oxygen are represented here: 2 Al(NO3)3 |
| 18 (2 × 3 × 3) |
| What are the units of molecular weight? |
| g/mol |
| What step of an equation must be complete before doing stoichiometry calculations? |
| The equation must be balanced first |
| What type of reaction is this? A + B → AB |
| Synthesis |
| What type of reaction is this? AB → A + B |
| Decomposition |
| What type of reaction is this? A + BC → B + AC |
| Single displacement |
| What type of reaction is this? AB + CD → AD + CB |
| Double displacement |
| What type of reaction is this? H(A) + (B)OH → AB + H2O |
| Neutralization |
| What type of reaction is this? CxHy + O2 → CO2 + H2O |
| Combustion |
| What type of reaction is this? Solid bromine reacts with sodium iodide |
| Single displacement |
| What type of reaction is this? Heating solid iron in the presence of oxygen. |
| Synthesis |
| What does a higher ranking mean on the reactivity series table? |
| Elements with a higher ranking can displace elements with a lower ranking |
| What is atomic mass? |
| The number of protons plus neutrons |
| What is an average atomic mass? |
| This is an average of all the different isotopes of that element found in nature. (The atomic mass on your periodic table is a decimal number because it is an average of different percentages of all the different isotopes) |
| If the percent abundance of an imaginary element is 50% 1.5g/mol and 50% 2.0g/mol, then what is the average atomic mass? |
| 1.75 g/mol |
| What is the magnitude of Avogadro's number and what does this number represent? |
| 6.02 × 1023 this number represents the quantity of particles (atoms, molecules, etc) found in 1.0 mol of a substance. (this number is so large that one mole of loonies would reach the Moon). |
| How is the mass calculate from the amount of moles and the molecular weight? |
| mass = (moles)(molecular weight) |
| How would you calculate the amount of moles given the number of particles of a substance? |
| Distinguish between an empirical and molecular formula. |
| An empirical formula is the lowest ratio of the elements in a compound while the molecular formula shows the ratio in its true, unreduced form. |
| Can the molecular formula be the same as the empirical formula? |
| Yes, if the molecular formula is the same as the empirical formula. |
| What two values are required to calculate the molecular formula of a compound? |
| Empirical formula and the molecular weight of the compound. |
| How is the Limiting Reactant calculated? |
| Divide the amount of moles present by the stoichiometric coefficient. The lowest magnitude is limiting. |
| What is the Limiting Reactant and the excess reactant (reagent)? |
| The Limiting Reactant is the molecule that gets consumed first. Excess reactant (reagent) are left over after the reaction is complete when the limiting is used fully. |
| What is the difference between actual yield and theoretical yield? |
| An actual yield is determined experimentally (in a lab) while the theoretical yield is calculated with a balanced equation and stoichiometry. |
| How is percent yield calculated? |
Solutions and Solubility, Acid-Base
Solutions
Solutions are heterogeneous mixtures composed of a solute dissolved in a solvent.
Solution
Solutions are homogeneous mixtures composed of a solute dissolved in a solvent. Homogeneous mixtures are uniform - consistent throughout.
Note that both solutes and solvents can consist of any combination of gas, liquid, or solid phases.
Note that both solutes and solvents can consist of any combination of gas, liquid, or solid phases.
Solutions
An alloy is mixture of two or metal elements. The most common stainless steel alloy used is Stainless Steel 316, which contains a combination of the following elements: Fe, C, Cr, Ni, Mo, Mn, Si, and more. Stainless steel is a solution by definition.
Solution
An alloy is a homogeneous mixture of elements, therefore it is a (solid) solution.
Ions in Solution
Electrolyte solutions contain aqueous ions that allow the conduction of electricity.
Solution
Soluble ionic compounds form electrolytes that can propagate charge movement, conducting electricity.
Which of the following is an electrolyte solution?
Solution
Only ionic compounds that dissolve in water form electrolyte solutions.
Hydrogen Bonding
A hydrogen bond is:
Solution
Hydrogen 'bond' is a bit of a misnomer, as it isn't actually a bond by definition, it is more of an electrostatic 'attraction'.
Hydrogen bonding can occur between molecules that contain Hydrogen (H) covalently bonded to any of Nitrogen (N), Oxygen(O), or Fluorine (F). Furthermore, hydrogen bonding can occur between molecules with any of N-H, O-H, F-H bonds and the lone pairs on N, O, or F.
Solution
True
Hydrogen Bonding
Which of the following pair of molecules cannot form hydrogen bonds?
Solution
H2O and CO2
The O-H bond in H2O can hydrogen bond to the lone pair of electrons on the oxygen in CO2 (even when the oxygen in CO2 is not bonded to hydrogen.)
CN: and H2O
The lone pair on a nitrogen atom acts as a hydrogen bond acceptor to the O-H bond in H2O.
NH3 and H2O
The N-H bond in NH3 and the O-H bond in H2O can clearly form strong hydrogen bonds.
CF4 and NH3
Surprisingly, the partial negative lone pair on fluorine in C-F: can hydrogen bond to the partial positive hydrogen in the H-N bond in NH3.
CN: and CF4
CN: and CF4 can only form hydrogen bonds with other molecules containing either O-H, N-H, or F-H bonds.
The O-H bond in H2O can hydrogen bond to the lone pair of electrons on the oxygen in CO2 (even when the oxygen in CO2 is not bonded to hydrogen.)
CN: and H2O
The lone pair on a nitrogen atom acts as a hydrogen bond acceptor to the O-H bond in H2O.
NH3 and H2O
The N-H bond in NH3 and the O-H bond in H2O can clearly form strong hydrogen bonds.
CF4 and NH3
Surprisingly, the partial negative lone pair on fluorine in C-F: can hydrogen bond to the partial positive hydrogen in the H-N bond in NH3.
CN: and CF4
CN: and CF4 can only form hydrogen bonds with other molecules containing either O-H, N-H, or F-H bonds.
Solubility of Compounds
Nonpolar compounds cannot dissolve in water because London dispersion forces are not as strong as dipole-dipole forces.
Solution
Polar compounds dissolve in water because of the dipole-dipole interactions. Compounds that can hydrogen bond are even more soluble. A molecule with neither hydrogen bonds nor dipole-dipole will be forced out of water because water prefers to bond/interact with other water molecules in the presence of nonpolar solutes.
Solubility of Compounds
Which of the following statements regarding the solubility of a compound is incorrect?
Solution
A diatomic molecule consisting of the same two atoms is nonpolar and would dissolve in a nonpolar solvent.
Percentage by Volume Concentration % V/V
Which of the following is the correct calculation of percentage by volume for 25mL of pure alcohol solute in 100mL of solution?
Solution
Percentage by Weight Concentration % W/V
Calculate the percentage weight by volume of 5.0 g of pure hydrochloric acid in 0.25 L of solution.
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
% (W/V)
Hint Unavailable
Convert L --> mL, by multiplying by 1.000.
0.25 L = 250 mL
0.25 L = 250 mL
Percentage Weight by Weight Concentration % W/W
Calculate the percentage weight by weight of 25 g of pure sugar in 500g of solution. (The w/w concentration is probably used on your toothpaste).
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
% (W/W)
Hint Unavailable
Summary of Concentrations (% V/V, % W/V, % W/W)
Determine the four INCORRECT combinations of units for calculating concentration (% V/V, % W/V, % W/W).
Solution
The 6 correct combinations are:
- g ÷ g
- g ÷ mL
- mL ÷ mL
- kg ÷ kg
- kg ÷ L
- L ÷ L
The 4 incorrect combinations are:
- g ÷ L
- g ÷ kg
- kg ÷ mL
- mL ÷ L
The little guys go with the little guys: g, mL...
The big guys go with the big guys: kg, L...
- g ÷ g
- g ÷ mL
- mL ÷ mL
- kg ÷ kg
- kg ÷ L
- L ÷ L
- g ÷ L
- g ÷ kg
- kg ÷ mL
- mL ÷ L
The big guys go with the big guys: kg, L...
Parts Per Million (PPM)
Which of the following would not equal 1.0 PPM?
Solution
Parts Per Million (PPM) is a form of Percentage Weight by Weight Concentration % W/W.
Note that PPM is only used for dilute solutions in which 1g ≈ 1mL.
Note that PPM is only used for dilute solutions in which 1g ≈ 1mL.
Parts Per Million (PPM)
Calculate the mass of solute dissolved in a 5.0L solution that is 0.3 parts per million (ppm).
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
mg
Hint Unavailable
Parts Per Million (PPM)
The local lake water is considered safe to swim in when the level of E. coli bacteria is 100 ppm. If this lake water was put in a bathtub with a 150L volume, what number of E. coli bacteria would be in a bathtub?
Solution
 Hint
Clear
Info
Incorrect Attempts:
CHECK
bacteria
Hint Unavailable
In this case the number of solute organisms is used instead of the mass.
Hint
Clear
Info
Incorrect Attempts:
CHECK
bacteria
Hint Unavailable
In this case the number of solute organisms is used instead of the mass.
Molarity
In solutions chemistry, the unit 'M' refers to
Solution
Molarity, mol/L
Calculating Molarity
1.0 mol/L of Na2O molecules contains 2.0 mol/L of Na+ ions.
Solution
One Na2O molecule dissolves into 2 Na+ molecules and 1 O2-. For every 1.0 mol/L of Na2O molecules, there are 2.0 mol/L of Na+ ions and 1.0 mol/L of O2- ions.
The concentration of chloride anions (Cl-) in 1.0 mol of MgCl2, dissolved in 1.0L of water is 2 mol/L
Solution
MgCl2 dissociates into 2 chloride (Cl-) ions:
1.0 mol of MgCl2 dissolved in 1.0L of water = 1 mol/L MgCl2.
Since there are every 1 MgCl2 forms 2 Cl- ions, then the molarity of chloride anions (Cl-)
= 2(1 mol/L)
= 2 mol/L
Since there are every 1 MgCl2 forms 2 Cl- ions, then the molarity of chloride anions (Cl-) = 2(1 mol/L)
= 2 mol/L
Calculating Molarity
Determine the molarity (molar concentration) of 1.0 mol NaCl dissolved in 250mL of water.
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
mol/L
Hint Unavailable
Although 1 mol NaCl dissociates into 2 mols of ions: Na+ + Cl-, the molar concentration uses the mols of the solute, or compound instead of the total ion equivalent.
Calculating Molarity
Determine the mass of potassium nitrate (KNO3) required to make 1,250mL of a 0.50M solution.
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
g
Hint Unavailable
Calculating Molarity
120mL of 4.5 mol/L potassium fluoride (KF) is added to 100mL of pure water. Determine the new molarity of the solution.
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
mol/L
Hint Unavailable
Calculating Molarity
Determine the final concentration when 3.0 L of 0.30 M NaCl is mixed with 1.0 L of 1.5 M NaCl?
Solution
Cf =
Hint
Clear
Info
Incorrect Attempts:
CHECK
mol/L
Hint Unavailable
0.4 mol of NaCl is mixed with 0.3 mol of AlCl3 in 2.0L of distilled water. Calculate the final concentration of chloride ions in solution [Cl-].
Solution
Cf =
Hint
Clear
Info
Incorrect Attempts:
CHECK
M
Hint Unavailable
NaCl and AlCl3 dissociate (break apart) into cations and anions in solution.
NaCl has 1 [Cl-] per molecule.
= 1(0.4 mol) of Cl- per molecule = 0.4 mol
AlCl3 has 3 [Cl-] per molecule.
= 3(0.3 mol) of Cl- per molecule = 0.9 mol
NaCl has 1 [Cl-] per molecule.
= 1(0.4 mol) of Cl- per molecule = 0.4 mol
AlCl3 has 3 [Cl-] per molecule.
= 3(0.3 mol) of Cl- per molecule = 0.9 mol
Dilution
Dilutions involve the addition of pure water to a solution.
Diluting a 2.0 mol/L solution of NaCl with distilled water will change the amount of moles of NaCl.
Solution
Dilutions with pure water do not change the solvent amount (moles). Moles are constant.
However, the concentration does change.
However, the concentration does change.
25mL of a 4mol/L solution is used to prepare a 2.5mol/L solution. Determine the final volume in milliliters.
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
mL
Hint Unavailable
Dilution
45mL of stock solution is taken from a 2.0L supply and used to make 100mL of 0.5mol/L solution. Determine the concentration of the stock solution.
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
mol/L
Hint Unavailable
Givens:
V1 = 45mL = 0.045L
V2 = 100mL = 0.1L
C2 = 0.5mol/L
Find C1
V1 = 45mL = 0.045L
V2 = 100mL = 0.1L
C2 = 0.5mol/L
Find C1
Gas Solubility
Gases cannot dissolve in liquids.
Solution
Many gases dissolve in water, for instance the carbon dioxide in soda. Also swimming pools contain a small amount of chlorine gas.
Solubility Curve
Describe the slope of NH3 (g) and AgNO3 (aq) on a solubility curve of concentration (g/100 mL) over temperature (˚C).
Solution
NH3 is a gas at room temperature. The solubility of gases decreases with increasing temperature; therefore the slope is negative.
AgNO3 is a solid, ionic compound that is soluble is water. The solubility of ionic compounds increases with increasing temperature; therefore the slope is positive.
AgNO3 is a solid, ionic compound that is soluble is water. The solubility of ionic compounds increases with increasing temperature; therefore the slope is positive.
Solubility
Which of the following statements is incorrect?
Solution
Polar liquids cannot dissociate, but they can dissolve. Solid ionic compounds dissociate, or break apart in solution.
Solubility
Determine the solubility of each ionic compound in water, as soluble, or insoluble.
| Order (Ranking) | Cation | Anion | Solubility | Exceptions |
| 1 (Highest) | Li+, Na+, K+, NH4+ | NO3-, ClO3-, CH3COO- | Soluble | Insoluble: Ca(ClO3)2 |
| 2 | Ag+, Hg2+, Pb+ | OH-, PO43-, CO32-, O2-, S2-, | Insoluble | Soluble: BaO, Ba(OH)2, and Group 2 Sulfides |
| 3 | Cl-, Br-, I- | Soluble | ||
| 4 | Ca2+, Sr2+, Ba2+ | Insoluble | ||
| 5 (Lowest) | Mg2+, Al3+, Fe2+, Fe3+, Cu2+, Zn2+ | SO42- | Soluble |
Potassium Hydroxide, KOH
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
Soluble
Due to potassium: KOH
Due to potassium: KOH
Calcium Carbonate, CaCO3
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
Insoluble
Due to Carbonate: CaCO3
Due to Carbonate: CaCO3
Calcium Nitrate, Ca(NO3)2
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
Soluble
Due to Nitrate: Ca(NO3)2
Due to Nitrate: Ca(NO3)2
Barium Chloride, BaCl2
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
Soluble
Due to Chloride: BaCl2
Due to Chloride: BaCl2
Silver Chloride, AgCl
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
Insoluble
Due to Silver: AgCl
Due to Silver: AgCl
Mercury(II) Sulfate/Sulphate, HgSO4
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
Insoluble
Due to Mercury: HgSO4
Due to Mercury: HgSO4
Magnesium Oxide, MgO
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
Insoluble
Due to Oxide: MgO
Due to Oxide: MgO
Calcium Sulfate/Sulphate, CaSO4
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
Insoluble
Due to Calcium, CaSO4
Due to Calcium, CaSO4
Lead(I) Nitrate, PbNO3
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
Soluble
Due to Nitrate: PbNO3
Lead, Pb, is a very insoluble substance, however when combined with Nitrate, NO3, which is the most soluble anion, the compound can dissociate/dissolve in water.
Due to Nitrate: PbNO3
Lead, Pb, is a very insoluble substance, however when combined with Nitrate, NO3, which is the most soluble anion, the compound can dissociate/dissolve in water.
Strontium Acetate, Sr(CH3COO)2
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
Soluble
Due to Acetate: Sr(CH3COO)2
Due to Acetate: Sr(CH3COO)2
Iron(II) Phosphate, Fe3(PO4)2
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
Insoluble
Due to Phosphate: Fe3(PO4)2
Due to Phosphate: Fe3(PO4)2
* Barium Oxide, BaO
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
Soluble
*Exception.
Ba2+ and O22 are both insoluble oxides, except when combined, the compound is soluble!
*Exception.
Ba2+ and O22 are both insoluble oxides, except when combined, the compound is soluble!
Lead(I) Chloride, PbCl
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
Insoluble
Due to Lead: PbCl
Due to Lead: PbCl
* Beryllium Sulfide, BeS
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
Soluble
*Exception.
Sulfides, S2- are insoluble, except when combined with the group 2 cations, alkaline earth metals.
*Exception.
Sulfides, S2- are insoluble, except when combined with the group 2 cations, alkaline earth metals.
Solubility
Hardwater contains Ca2+ ions dissolved in solution. Ions are taken out of solution by precipitation into solids. Which of the following compounds would be best at lowering the calcium ion concentration in water.
Solution
NaSO4 would precipitate the Ca2+ because calcium is more reactive and will displace the sodium ions.
The sulfate/sulphate becomes insoluble when bound to calcium - this precipitates the calcium out of solution, lowering its concentration. None of the other compounds would precipitate calcium.
The sulfate/sulphate becomes insoluble when bound to calcium - this precipitates the calcium out of solution, lowering its concentration. None of the other compounds would precipitate calcium.
Net Ionic Equation
Determine the net ionic equation for the reaction of potassium carbonate and aluminum nitrate. [Side note: the product of the reaction is not common in nature, meaning it usually doesn't form spontaneously, naturally.]
Solution
FIRST: Separate the ions of all soluble compounds, leaving the solid, liquid, and gaseous compounds.
SECOND: Cancel ions on the left and right side, leaving the net ionic equation.
SECOND: Cancel ions on the left and right side, leaving the net ionic equation.
Solubility of Relative Unknowns
Four test tubes are filled with the following four ions: Na+, Ca2+, SO42+, and OH-. However, it is not known which ion is in which test tube. Four different mixtures are prepared from the test tubes, and the observations are recorded in the table below. Using your understanding of solubility, determine which ion is in each test tube. Solution
| Mixtures | Test Tube 2 | Test Tube 4 |
| Test Tube 1: | Soluble | Soluble |
| Test Tube 3: | Insoluble | Insoluble |
Test Tube 2: OH-
Test Tube 3: Ca2+
Test Tube 4: SO42+
(Note that it could be switched where Test Tube 2 is SO42+ and Test Tube 4 is OH-.)
Molarity Calculation with Volume
Determine the volume of 0.75 M KCl needed to react completely with 100.0 mL of 0.50M NaNO3.
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
mL
Hint Unavailable
Molarity Calculation with Volume
If 0.50 L of 1.500 mol/L Na2O is mixed with excess AgClO3, what mass of precipitate will form?
Svante Arrhenius 1884
Arrhenius defined acids and bases as
Solution
According to Arrhenius,
Acids are proton (H+) donors, and bases are hydroxide (OH-) donors
(Arrhenius' definitions become outdated when Brønsted and Lowry offered their definitions, and later Lewis...)
Acids are proton (H+) donors, and bases are hydroxide (OH-) donors
(Arrhenius' definitions become outdated when Brønsted and Lowry offered their definitions, and later Lewis...)
Johannes Brønsted and Thomas Lowry 1923
Brønsted and Lowry defined acids and bases as
Solution
According to Brønsted and Lowry,
Acids are proton (H+) donors, and bases are proton (H+) acceptors
(Brønsted and Lowry's definition was more general than Arrhenius' and encompassed more circumstances)
Acids are proton (H+) donors, and bases are proton (H+) acceptors
(Brønsted and Lowry's definition was more general than Arrhenius' and encompassed more circumstances)
Brønsted Amphoteric
A water molecule can act as both a Brønsted acid and a Brønsted base.
Solution
Water can donate a proton to become OH-. Water can also accept a proton to become H3O+. This depends on what else is there with the water. For example...
Arrhenius, Brønsted-Lowry
Ammonia (NH3) violates which definition of a base?
Solution
Ammonia does not contain hydroxide (OH-) ions.
For example in pure water, Ammonia, NH3 is a Brønsted-Lowry base because it is a proton acceptor, but is not a hydroxide donor according to Arrhenius' definition. (Arrhenius defined bases as hydroxide donors).
As you can see the base, ammonia is a proton acceptor (Brønsted-Lowry base) but does not fall under the Arrhenius category of a base (hydroxide donor).
For example in pure water, Ammonia, NH3 is a Brønsted-Lowry base because it is a proton acceptor, but is not a hydroxide donor according to Arrhenius' definition. (Arrhenius defined bases as hydroxide donors). As you can see the base, ammonia is a proton acceptor (Brønsted-Lowry base) but does not fall under the Arrhenius category of a base (hydroxide donor).
Brønsted-Lowry Base Strength
Which of the following Brønsted bases is the strongest?
Solution
A Brønsted base is a proton acceptor. Fluoride ions are more electronegative and have a smaller ionic radius.
Furthermore, the conjugate acid HF is the weakest acid. Remember that the conjugate base of a weak acid is a strong base.
Furthermore, the conjugate acid HF is the weakest acid. Remember that the conjugate base of a weak acid is a strong base.
Acid Base Neutralization
Hydrochloric acid neutralizes sodium hydroxide base in the following reaction. What are the products of this reaction?
Solution
Acid + Base ↔ Salt + Water...
Identifying Acids and Bases
Which of the following mixtures would result in a neutralization reaction? Solution
| Mixture | Compound 1 | Compound 2 |
| A | HCl | HNO3 |
| B | K2SO4 | H2O |
| C | OH- | H2CO3 |
| D | CH3COO- | NaOH |
Acid = H2CO3
Base = OH-
Acids and Bases
Which of the following statements about acids and bases is false?
pH Units
What are the units used for pH?
Calculating pH
Which sample has an incorrect pH calculation in the following table? Solution
| Sample | [H+(aq)] | pH |
| A | 1.0 × 10—13 | 13 |
| B | 1.0 × 10—2 | 2 |
| C | 1.0 × 10—8 | 8 |
| D | 1.0 × 100 | 1 |
| E | 1.0 × 10—5 | 5 |
[1.0 × 100] has a pH = 0
Calculating pH
Calculate the pH of a solution with [H+(aq)] = 2.8 × 10-3 mol/L.
Which of the following quantities of H+ would make an acidic 5.0 L solution?
Solution
Note: you must divide the moles by the 5.0L volume to obtain the concentration in mol/L.
The acidic solution has a pH less than 7 and a [H+] > 1.0 × 10-7 mol/L.
The acidic solution has a pH less than 7 and a [H+] > 1.0 × 10-7 mol/L.
Calculating pH
Calculate the pH of a solution of 0.150 mol/L of H2SO4, a strong acid.
Solution
pH =
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
H2SO4 contains 2 protons per molecule.
So, 2 H3O+ molecules will be made from one molecule of H2SO4.
So, 2 H3O+ molecules will be made from one molecule of H2SO4.
Conjugate Pair of Acids and Bases
Which of the following is a conjugate acid-base pair?
Solution
The carbonic acid H2CO3 dissociates into the bicarbonate anion, which forms a salt with the available sodium ions, as sodium bicarbonate: NaHCO3
Conjugate Pair of Acids and Bases
Which of the following is a conjugate acid-base pair?
Acid Base Titrations
The following table shows the ratio of strong acid and strong base required to neutralize. Determine which pair has an incorrect ratio. Solution
| Acid | Base |
| 2 H3PO4 | 3 Mg(OH)2 |
| 1 H2SO3 | 2 NaOH |
| 1 HCl | 1 KOH |
| 1 H3O+ | 3 OH- |
| 2 HNO2 | 1 Ba(OH)2 |
1 H3O+ and 1 OH-
Titrations and Indicators
Universal indicators are used to test a wide range of pH.
 |
The point at which the indicator starts to change color is the:
Solution
As the pH of solution changes, the indicator will change color at the endpoint when the specific pH for that indicator is reached.
(Note that many metallic ions make different color solutions and this is not an endpoint).
(Note that many metallic ions make different color solutions and this is not an endpoint).
What is the equivalence point in a titration?
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
The equivalence point is the point at which the amount of moles of the acid equals the moles of the base to completely neutralize the other.
Some titrations with polyprotic acids or polyhydroxic bases.
Some titrations with polyprotic acids or polyhydroxic bases.
Acid Base Titrations
What volume of 2.0 M H3PO4 will neutralize 500 mL of 1.5 M NaOH? Write the neutralization equation.
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
L
Hint Unavailable
Neutralization occurs when moles of acid = moles of base:
Acid Base Titrations Lab
A chemistry student has titrated a strong base using a strong acid. A 0.5M hydrochloric acid (HCl) was slowly dripped one drop at a time from a buret into a beaker containing an unknown concentration of 50mL of Mg(OH)2. The volume of acid that changed the colour of the indicator in the unknown sample was recorded in the table. Find the unknown concentration of base. Solution
| Trial: | 1 | 2 | 3 |
| Initial buret reading (mL): | 42.0 | 30.4 | 19.0 |
| Final buret reading (mL): | 30.4 | 19.0 | 6.9 |
| Volume of HCl added: | 11.6 | 11.4 | 12.1 |
Titration Curves
The following titration curve represents
Solution
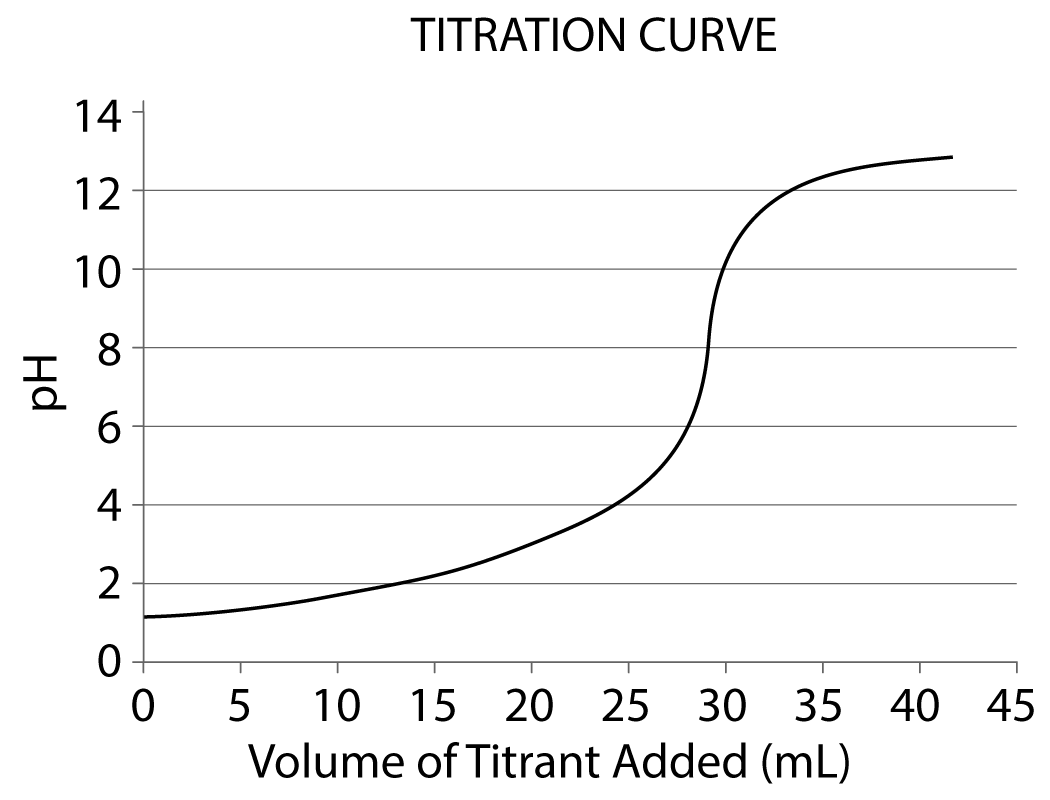
At the equivalence point, the moles of acid is equal to the moles of base. The equivalence point of the titration is > 7 as you can see on the graph. An acidic equivalence point indicates a stronger acid, a basic equivalence point indicates a stronger base, and a neutral equivalence point indicates equal strength of acid and base.
Solutions Units
1 mmol equals
Solution
Milli, 'm', represents 1/1000th
pH and pOH
There is a constant relationship between pH and pOH.
The following equations always apply to acids and bases.
Solution
1 × 10-14 = [OH-][H+]
14 = pH + pOH
True. These equations are used often, see below.
Determine the pH of a 0.5 M solution of NaOH
Solution
pH =
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
Determine pOH based on the concentration [OH-] = 0.5 M
pH + pOH = 14
Concentration Combinations
A 1.1 L of a 0.6 M solution of H3O+ is added to a 2.5 M stock solution of H3O+. If the total concentration is 1.5 M, determine the volume of the stock solution.
Solution
V =
Hint
Clear
Info
Incorrect Attempts:
CHECK
L
Hint Unavailable
Let the volume of the stock solution be Vs
Acids
Calculate the pH assuming the acid completely dissociates into PO43-.
Solution
pH =
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
H2PO4 → 3H+ + PO43-
Convert mass to mol
Stoichiometry: 1H2PO4 → 3H+
Convert mol H+ to concentration first, [H3O+]
Calculate pH based on concentration, [H3O+]
Acid Base Reaction pH
80mL of a 2.5M solution of sulfuric acid is added to 110mL of a 1.1mol/L solution of sodium hydroxide.
Determine the total number of moles of protons (H+) and hydroxides (OH-) in solution before neutralization occurs.
Solution
Hint
Clear
Info
mol H+ =
mol OH- =
Incorrect Attempts:
CHECK
Hint Unavailable
There are total 0.4 mol of protons (H+), and 0.121 mol of hydroxides (OH-) in solution, before neutralization.
mol OH- =
Determine the limiting reagent and the excess amount of moles of protons or hydroxides after neutralization.
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
mol
Hint Unavailable
The NaOH is limiting because the lower amount of total moles.
Neutralization = 1 H+ + 1 OH- = 1 H2O
Neutralization = 1 H+ + 1 OH- = 1 H2O
Calculate the pH
Solution
pH =
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
first need to calculate the concentration of [H+]
(Use total Volume)
then calculate pH
(Use total Volume) then calculate pH
Flashcards: Solutions and Solubility
| What is the difference between a homogeneous and heterogeneous mixture? |
| Homogeneous mixtures are uniform/consistent throughout and the components are indistinguishable. The components of heterogeneous mixtures can be distinguished visually. |
| Explain why a metal alloy is or is not a solution |
| An alloy is a uniform mixture of two or more metals. It is possible to have solid solutions, liquid solutions, and gas solutions. |
| What is an electrolyte? |
| An electrolyte is a solution composed of dissolved (aqueous) ions that allow the solution to conduct electricity. |
| Explain how the saturation point of a solution can be changed. |
| By changing the temperature of the solution. (For a solid, increasing the temperature increases the saturation point while decreasing the temperature decreases the saturation point. The opposite is true for gases.) |
| Explain why sugar water is or is not an electrolyte solution. |
| Sugar is not an electrolyte solution because it is not ions dissolved in a solvent — electrolytes are made only of aqueous ions. Sugar is a covalent/molecular compound and does not dissolve as ions in solution. |
| What are the aqueous ions formed when sulfuric acid (H2SO4) dissolves? |
| H+ and SO42- |
| What is the difference between dissolving and dissociation? |
| Dissociation is any chemical splitting of a molecule into its smaller parts, while dissolving is the chemical splitting of a molecule due to the presence of a liquid such as water. |
| Correct the following statement: Hydrogen bonding can occur whenever there are hydrogen bonds in a molecule. |
| Hydrogen bonding can occur when the hydrogen is bonded to any one of three highly electronegative atoms: nitrogen, oxygen, or fluorine. The recipient of the hydrogen bond must have a partial negative dipole (like the oxygen in OH), or a negative charge like a lone pair of electrons. |
| What is parts per million (PPM)? |
| This is a density, calculated in several ways as either: grams particles ÷ million grams solute grams particles ÷ million mL solute grams particles ÷ thousand L solute ... |
| What is molarity (M)? |
| The amount of solute per in a certain volume of solution. mole/Liter |
| What is the concentration of chloride ions in a 3M solution of MgCl2? |
| 6M |
| How is volume calculated, given concentration and moles? |
| How does temperature affect the solubility of a gas in water? |
| The relationship is inverse. For example, the solubility of gases decreases with increasing temperature. |
| Predict the solubility of any cation bonded to nitrate (NO3-) |
| Soluble (nitrate is one of the most soluble ions known: any ionic compound containing nitrate is soluble in water) |
| State the net ionic equation of |
(net ionic equations only include the ions that contribute to making the solid product) |
| What is a spectator ion? |
| Spectator ions are ions that are present on both sides of the ion equation and do not contribute to make the solid product. |
Gases and Atmospheric Chemistry
Physical Properties of Gases
Which property of substances is considered responsible for determining the physical states of substances with similar composition as solid, liquid, or gas?
Solution
Intermolecular forces: dipole-dipole, hydrogen bonding, van der waals... determine physical state.
Physical state is also based on molecular weight (g/mol) to a lesser extent:
E.g.) Heavier compounds C20H42 are more likely solids compared to lighter compounds with the same atoms C2H6.
Physical state is also based on intramolecular force to a lesser extent:
E.g.) Substances that contain ionic bonds NaCl are more likely solids than different substances with only covalent bonds NH3.
(Temperature/heat also affects physical state, but is not considered a property of a substance.)
Physical state is also based on molecular weight (g/mol) to a lesser extent:
E.g.) Heavier compounds C20H42 are more likely solids compared to lighter compounds with the same atoms C2H6.
Physical state is also based on intramolecular force to a lesser extent:
E.g.) Substances that contain ionic bonds NaCl are more likely solids than different substances with only covalent bonds NH3.
(Temperature/heat also affects physical state, but is not considered a property of a substance.)
Intermolecular forces are the only factor that determines the physical state of a substance.
Solution
Temperature is proportional to kinetic energy. The physical state also depends on the kinetic energy of the molecules. Molecules with higher kinetic energies tend to have higher volumes, vapor pressure, and other things.
Other factors are temperature, and identity of the substance.
Other factors are temperature, and identity of the substance.
Physical Properties of Gases
Why are gases compressible?
Solution
Unlike solids or liquids, gas molecules can be squeezed closer together because they are spaced relatively far apart to begin with. The Kinetic Molecular Theory says the space occupied by the gas molecules is negligible compared to the total volume.
Physical Properties of Gases
100g of a gas occupies much more volume than 100g of the same solid. What useful application could this property be used for?
Solution
The instantaneous expansion of airbags is due to a highly reactive mass of solid turning into a gas very quickly. The gas occupies a much larger volume than the solid state and inflates the airbag (quickly).
Types of Intermolecular Forces in Gases
Determine the types of intermolecular forces that exist in the following gases.
Argon
Solution
Argon is an inert Noble Gas with no polarity so it has London dispersion forces only.
O2
Solution
Diatomic oxygen, O2 has no polarity so it has London dispersion forces only.
NH3
Solution
Ammonia has both London dispersion and dipole-dipole forces, this makes it the combined van der Waals, which includes both London dispersion and dipole-dipole forces.
Kinetic Molecular Theory
This physical state has the highest internal kinetic energy for a given substance.
Standard Temperature and Pressure (STP)
Which of the following is the standard temperature and pressure (STP)?
Solution
Not to be confused with the newer standard atmospheric temperature and pressure (SATP), which is 100kPa and 25˚C.
Units of Pressure
Determine the equivalent value of Pascals (Pa) in 100 kPa.
Solution
1 kPa = 1,000 Pa
Units of Pressure
A noble gas is stored in a laboratory tank at 5 atm.
Determine the equivalent pressure in kPa, given 1 atm = 101.325 kPa
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
kPa
Hint Unavailable
Convert this the previous value in kPa to torr, given 101.325 kPa = 760 torr.
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
torr
Hint Unavailable
Gas Laws
Which of the following is Charles' Law?
Solution
Charles' Law:
Gay-Lussac's Law:
Avogadro's Law:
Boyle's Law: P1V1 = P2V2
Ideal Gas Law: PV = nRT
Gay-Lussac's Law:
Avogadro's Law:
Boyle's Law: P1V1 = P2V2
Ideal Gas Law: PV = nRT
When using Charles' law, it does not matter what version of units of temperature are used.
Solution
Temperature units must be converted to Kelvin. ˚C cannot be used in the Charles' law equation. (Note that any unit can be used for volume, commonly dm3, or L)
Charles' law can be written as either of the following:
Solution
Due to the law of proportionality, it does not matter which of these two orders the law is written in.
Which version of the following units must be used in Charles', Boyle's, or Gay-Lussac's law?
Solution
The units of pressure and volume do not matter (as long as they are consistent).
The only unit that must be used is Kelvin for Temperature.
The only unit that must be used is Kelvin for Temperature.
Charles' Law
In the following relationship, V is inversely proportional to T.
Solution
For example, with a constant value for 'k', increasing temperature by a factor of two, must coincide with the same increase in volume:
V is directly proportional to T
Converting Kelvin and Celsius
Convert 273K to Celsius:
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
˚C
Hint Unavailable
Celsius = Kelvin - 273
Kelvin = 273K - 273
= 0˚C
Kelvin = 273K - 273
= 0˚C
Gas Laws
Solve the following problems.
What must be maintained constant in Boyle's Law?
Solution
PV = k
Boyle's Law compares Volume and Pressure. The product of volume and pressure is constant because their relationship is inversely proportional (as one increases, the other decreases).
(Note that pressure and amount of gas must be kept constant to use Charles' Law.)
(Note that pressure and amount of gas must be kept constant to use Charles' Law.)
A 10.0L gas canister contains air at 3atm. If 2.0L of air is added to the canister while temperature and amount of the gas is constant, what is the new pressure?
Solution
P =
Hint
Clear
Info
Incorrect Attempts:
CHECK
atm
Hint Unavailable
Givens:
P1 = 3 atm
V1 = 10.0 L
V2 = Total Volume = 10.0 L + 2.0 L
Find P2.
Use Boyle's Law
P1 = 3 atm
V1 = 10.0 L
V2 = Total Volume = 10.0 L + 2.0 L
Find P2.
Use Boyle's Law
A constant amount of gas in an isobaric chamber is cooled from 300˚C to 100˚C. If the starting volume was 5.0L, find the new volume.
Solution
V =
Hint
Clear
Info
Incorrect Attempts:
CHECK
L
Hint Unavailable
Givens:
T1 = 300˚C
T2 = 100˚C
V1 = 5.0L
Find V2.
Use Charles' Law
T1 = 300˚C
T2 = 100˚C
V1 = 5.0L
Find V2.
Use Charles' Law
The Combined Gas Law
Given the following three gas laws, determine the correct order of the combined gas law based on the proportionality of the given laws:
Boyle's Law: P1V1 = P2V2
Charles' Law:
Gay-Lussac's Law:
The Combined Gas Law
A gas canister contains 35mL of N2 (g) at 580mmHg and 30˚C. What volume would the gas occupy at STP?
Solution
V =
Hint
Clear
Info
Incorrect Attempts:
CHECK
mL
Hint Unavailable
Givens:
STP is 0˚C (273K) and 101.3kPa
P2 = 101.3kPa = 760mmHg
T2 = 273K
V1 = 35mL
P1 = 580mmHg
T1 = 30˚C + 273K = 303K
Make sure to convert units to be consistent.
Temperature must be in K.
STP is 0˚C (273K) and 101.3kPa
P2 = 101.3kPa = 760mmHg
T2 = 273K
V1 = 35mL
P1 = 580mmHg
T1 = 30˚C + 273K = 303K
Make sure to convert units to be consistent.
Temperature must be in K.
Gas Law Relationships Curves
Determine the correct relationship in the following graphs, assuming other factors are kept constant and the gas behaves ideally.
Solution
Notice that P-T and V-T have a linear relationship:
The relationship between P and T is also linear :

These relationships can also be explained with the combined gas law.
(n-V has a linear relationship where V is a horizontal line, i.e. the V is due to the space between the gases since the gas particles have no volume).

These relationships can also be explained with the combined gas law.
(n-V has a linear relationship where V is a horizontal line, i.e. the V is due to the space between the gases since the gas particles have no volume).
Gas Law Relationships
Determine the slope on a graph of n versus V, assuming other factors are kept constant and the gas behaves ideally.
Solution
Avogadro's Law:
P (↑V) = (↑n) R T
Moles (n) and Volume (V) are directly proportional, as one increases, the other increases. A graph with n on the x-axis and V on the y-axis would have a linear, positive slope.
Gas Law Relationships
Describe the relationship on the graph between V (on the x-axis) versus PV (on the y-axis).
Solution
P V = k
↑P ↓V = ~k
Boyle's Law states that at constant temperature and amount of gas, the product of volume and pressure is constant because their relationship is inversely proportional (as one increases, the other decreases). 'k' is constant.
A V—PV graph would appear as a horizontal line (with a slope of zero). As V increases, P decreases by the inverse factor so PV is flat.
A V—PV graph would appear as a horizontal line (with a slope of zero). As V increases, P decreases by the inverse factor so PV is flat.
Ideal Gas Theory
A scientist is calculating the temperature of an ideal gas. She measures the volume and the amount of moles of the sample. To determine the temperature, what other measurement must the scientist make? (R = 8.31)
Solution
PV = nRT
Ideal Gas Theory
Ideal gas particles (atoms, molecules) have zero volume.
Solution
For the volume to be based on the intermolecular interactions, the molecules themselves are assumed to occupy zero volume.
Increasing the amount of moles (n) of an ideal gas increases the volume of that gas.
Solution
While it is true that ideal gas particles (atoms, molecules) are considered to have zero volume, it is the space between the gas particles that does occupy a volume and that's what counts.
The Ideal Gas Law
The ideal gas law contains relationships derived from which of the following gas laws?
Solution
Ideal Gas Law: PV = nRT
Charles' Law:
Gay-Lussac's Law:
Avogadro's Law:
Boyle's Law: P1V1 = P2V2
Charles' Law:
Gay-Lussac's Law:
Avogadro's Law:
Boyle's Law: P1V1 = P2V2
The Ideal Gas Law
The ideal gas constant R = 8.31, must use which units for Pressure, Volume, and Temperature?
Solution
If units are not given in kPa, L, and K then they must be converted to these units before used in the equation.
R = 8.31 kPa·L/mol·K
R = 8.31 kPa·L/mol·K
The Ideal Gas Law
Given the following values, determine the correct equation to find the amount of moles. (R = 8.13) Solution
| Pressure: | 101,325 Pa |
| Volume: | 18,500 mL |
| Temperature: | 10 ˚C |
The Ideal Gas Law
10.0 L of carbon dioxide (CO2) gas is compressed into a canister at -20˚C and 4.71 atm.
Determine the amount of moles.
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
mol
Hint Unavailable
Determine the mass of this sample, given the molecular weight of carbon dioxide is 44 g/mol. Round to a whole number.
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
g
Hint Unavailable
MW CO2 = 44 g/mol.
Gas Laws and Equations
Glucose is produced in photosynthesis by the reaction below.
6CO2 + 6H2O → C6H12O6 + 6O2Calculate the volume of carbon dioxide that goes into producing 1.0 kg of glucose sugar at SATP.
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
L
Hint Unavailable
Givens:
T = 25˚C = 298 K (@SATP)
P = 100 kPa (@SATP)
R = 8.314 L·kPa/mol·K
m = 1.0 kg = 1000 g
Calculate n...
Then calculate V...
Convert mass to moles:
Use the stoichiometric ratio to get mol of reactant from mol or product:
T = 25˚C = 298 K (@SATP)
P = 100 kPa (@SATP)
R = 8.314 L·kPa/mol·K
m = 1.0 kg = 1000 g
Calculate n...
Then calculate V... Convert mass to moles: Use the stoichiometric ratio to get mol of reactant from mol or product:
Now with that amount from the previous part, calculate the new volume at STP.
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
L
Hint Unavailable
Givens:
T1 = 25˚C = 298 K (@SATP)
P1 = 100 kPa (@SATP)
V1 = 825.03 L
T2 = 0˚C = 273 K (@STP)
P2 = 100 kPa (@STP)
V2 = ??? L
Use the combined gas law... But see that the pressure is constant from SATP to STP, so use Charles' law instead...
T1 = 25˚C = 298 K (@SATP)
P1 = 100 kPa (@SATP)
V1 = 825.03 L
T2 = 0˚C = 273 K (@STP)
P2 = 100 kPa (@STP)
V2 = ??? L Use the combined gas law... But see that the pressure is constant from SATP to STP, so use Charles' law instead...
Now with that amount from the previous part, calculate the new volume if the temperature is kept constant and the pressure is changed to 836 torr.
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
L
Hint Unavailable
Givens:
P1 = 100 kPa (@STP)
V1 = 755.82 L
P2 = 836 torr
V2 = ??? L
First convert 'torr' to 'kPa'...
Use Boyle's law...
P1 = 100 kPa (@STP)
V1 = 755.82 L
P2 = 836 torr
V2 = ??? L First convert 'torr' to 'kPa'... Use Boyle's law...
Dalton's Law of Partial Pressure
For a given temperature and volume, which of the following gases has the greatest partial pressure?
Solution
For a given temperature and volume, partial pressure only depends on the amount of moles.
Three gases occupy a closed container. Gas 'A' has a partial pressure of 380 mmHg, while gas 'B' has a partial pressure of 202.65 kPa. If the total pressure is 4 atm, find the pressure of gas 'C', in atm.
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
atm
Hint Unavailable
First, convert all units to the same unit.
A closed container contains: 1.806 × 1024 molecules of CO2, 56.04 g of N2, and 1 mol of CH4. Calculate the total pressure in the closed container, in kPa, if the partial pressure of CH4 is 165.17 kPa.
Solution
PT =
Hint
Clear
Info
Incorrect Attempts:
CHECK
kPa
Hint Unavailable
Determine the total amount of moles and use the mole fraction to determine total pressure.
And,
Total moles:
Substitute the values into the mole fraction, partial pressure equation:
Partial Pressure and Ideal Gases
A 5.0 L insulated container contains the following mixture of gases at 0˚C and 2 atm:
| Gas | Percentage |
| N2 | 60% |
| O2 | 30% |
| C3H8 | 10% |
Determine the partial pressure of N2.
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
atm
Hint Unavailable
Dalton's Law of Partial Pressure: Ptotal = P1 + P2 + P3 + ...
In this question Ptotal = 2 atm
The partial pressure of N2 is 60% of 2 atm = 0.6(2 atm) = 1.2 atm
In this question Ptotal = 2 atm
The partial pressure of N2 is 60% of 2 atm = 0.6(2 atm) = 1.2 atm
Determine the amount of moles N2 in the mixture.
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
mol
Hint Unavailable
R = 8.13 kPa·L/mol·K
0˚C = 273K
0˚C = 273K
Application of the Ideal Gas Law
A 34.5g sample of an unknown gas is collected at room temperature (20˚C) and atmospheric pressure (101.325 kPa). The initial reading on a near-vacuum collection cylinder is 10mL and the final amount once filled is 18,040 mL.
Determine the amount of moles in the collection sample.
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
mol
Hint Unavailable
20 ˚C = 293 K
R = 8.314 kPa·L/mol·K
R = 8.314 kPa·L/mol·K
Determine the molecular weight of the sample.
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
g/mol
Hint Unavailable
n = 0.75 mol
m = 34.5 g
m = 34.5 g
Determine the identity of the gas.
Molecular weights are given to make it simpler for the student.
Gas Laws
Which of the following statements about gases is not a true fact about ideal gases?
Solution
Absolute zero (0.0 K) can never be reached so gas particles can never stop. [Edit: now possible.]
The volume occupied by a gas does not consist of the volume of the atoms or molecules—this volume comes from the molecules colliding and spreading apart from one another.
Since the gas volume comes from space between colliding gas particles, theoretically it cannot have any volume if the particles are not moving at 0K (although this cannot actually happen).
The state of an ideal gas is always gaseous when calculating, for instance the boiling point of ammonia (NH3) is -33.34 °C, but even at -50 °C, it would still be considered gaseous.
The volume occupied by a gas does not consist of the volume of the atoms or molecules—this volume comes from the molecules colliding and spreading apart from one another.
Since the gas volume comes from space between colliding gas particles, theoretically it cannot have any volume if the particles are not moving at 0K (although this cannot actually happen).
The state of an ideal gas is always gaseous when calculating, for instance the boiling point of ammonia (NH3) is -33.34 °C, but even at -50 °C, it would still be considered gaseous.
Gas Laws
Determine the outcome when each of the following changes are made to the following equation.
PV = nRTVolume will decrease by a factor of ¼ when pressure is doubled, temperature is halved, and moles are kept constant.
Solution
Therefore volume will decrease by a factor of ¼.
By what factor will temperature change when pressure is increased by a factor of 3, volume is decreased by a factor of ¾, and moles are increased by a factor of 2.
Solution
Therefore temperature will increase by a factor of .
Water Vapor Pressure
What is vapor pressure?
The Law of Combining Volumes
When reacted at the same temperature and pressure, gases will combine in the following ratios. Which one of the following ratios is incorrect? Solution
| Reactant 1 | Reactant 2 | Product | Ratio |
| H2 (g) | S (g) | H2S (g) | 1:1:1 |
| H2 (g) | Cl2 (g) | 2HCl (g) | 1:1:2 |
| 2H2 (g) | O2 (g) | 2H2O (g) | 2:2:2 |
| N2 (g) | 2O2 (g) | 2NO2 (g) | 1:2:2 |
| N2 (g) | 3H2 (g) | 2NH3 (g) | 1:3:2 |
| Reactant 1 | Reactant 2 | Product | Ratio |
| 2H2 (g) | O2 (g) | 2H2O (g) | 2:1:2 |
The Law of Combining Volumes
Which of the following proportions is incorrect in the formation of ammonia from its elemental compounds? Solution
| N2 (g) | + | 3H2 (g) | → | 2NH3 (g) | |
| A | 2.0 g | + | 6.0 g | → | 4.0 g |
| B | 1 mol | + | 3 mol | → | 2 mol |
| C | 1 L | + | 3 L | → | 2 L |
| D | 1 mL | + | 3 mL | → | 2 mL |
| E | 100 L | + | 300 L | → | 200 L |
Molar Volume
The volume of any gas at STP (0˚C, and 1 atm) is 22.4 L per 1 mole.
Solution
While 22.4 L per 1 mole is correct, molar volume is an approximation that only works for ideal gases rather than all gases.
Molar Volume
The molar volume (Vm) of an ideal gas at STP is 22.4 L/mol.
Determine the amount of moles in 3.2 L of carbon dioxide.
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
mol
Hint Unavailable
Determine the mass of this gas.
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
g
Hint Unavailable
molar mass of CO2 = 44 g/mol.
Molar Volume
Determine which gas contains the most molecules at STP.
Solution
3.0 L of 2NH3 (g)
3.0 L of 2 moles of NH3 (g) = 6.0L
This is the largest volume, which will yield the most moles (at 22.4L/mol).
This will become the most molecules (when multiplied by Avogadro's constant 6.02 × 1023 molecules/mol).
3.0 L of 2 moles of NH3 (g) = 6.0L
This is the largest volume, which will yield the most moles (at 22.4L/mol).
This will become the most molecules (when multiplied by Avogadro's constant 6.02 × 1023 molecules/mol).
Molar Volume
Determine the density of methane, CH4 (16 g/mol) at STP.
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
g/L
Hint Unavailable
Assuming methane behaves as an ideal gas, at STP it is 22.4 L/mol
You want to get mol to cancel, so you're left with g/L.
You want to get mol to cancel, so you're left with g/L.
Molar Volume
The molar volume (Vm) of an ideal gas at STP is 22.4 L/mol.
5 L of propane (C3H6) gas reacts with 6 L of oxygen. Determine the limiting reagent if these gases behave as ideal gases.
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
Hint Unavailable
No need to convert volume to moles if an ideal gas.
The lower number is the limiting reagent, therefore oxygen (O2) is limiting.
Determine the volume of carbon dioxide produced.
Solution
V =
Hint
Clear
Info
Incorrect Attempts:
CHECK
L
Hint Unavailable
Molar ratio of the limiting reagent, oxygen to the product, carbon dioxide is 9:6
Molar Volume
1.0L of hydrochloric acid is produced in the following reaction at STP.
Find the starting volume of hydrogen.
Solution
V =
Hint
Clear
Info
Incorrect Attempts:
CHECK
L
Hint Unavailable
Molar ratio (n) of HCl to H2 is 2:1
Fastest to use law of combining volumes, rather than convert to moles and back to volume.
0.5 L of hydrogen gas.
Fastest to use law of combining volumes, rather than convert to moles and back to volume. 0.5 L of hydrogen gas.
Find the amount of moles of hydrochloric acid if it behaves as an ideal gas.
Solution
Hint
Clear
Info
Incorrect Attempts:
CHECK
mol
Hint Unavailable
"Behaves as an ideal gas" means 22.4L/mol
0.04 mol of HCl.
Flashcards: Gases
| What is the most compressible phase? |
| Gas |
| Is this SATP or STP? 100 kPa and 25˚C |
| SATP |
| How many Pa are in a kPa? |
| 1,000 |
| What is the standard atmospheric pressure in kPa, atm, and mmHg |
| 101.3kPa, 1 atm, and 760mmHg |
| How does temperature affect kinetic energy of gas molecules? |
| Directly proportional |
| Who's law is this? |
| Charles' Law |
| Who's law is this? P1V1 = P2V2 |
| Boyle's Law |
| Who's law is this? |
| Gay-Lussac's Law |
| Who's law is this? |
| Avogadro's Law |
| Is 0K possible? |
| No |
| What is an ideal gas? |
| An assumption that gas particles do not have volume, except for the space between particles. There are no intermolecular forces between the gas molecules. Also that the gas is at a high temperature and low pressure. |
| What does Dalton's law of partial pressure mean for the volume of different gases in air? |
| The volume does not depend on the type of molecule, only on the percentage amount of the total volume. |
| A gas is cooled from 300˚C to 100˚C, what is the change in temperature in Kelvin? |
| 200K |
| What is the relationship of temperature versus volume in an ideal gas? |
Linear |
| What is the volume of 1 mol at STP? |
| 22.4L |
Hydrocarbons and Energy
Physical Property of Hydrocarbons
What types of bonds do hydrocarbons contain?
Solution
Hydrocarbons are non-polar and only have weak, temporary intermolecular forces, so the only type of intermolecular bonding is London dispersion.
It is not Van der Waals because this actually includes London as well as dipole-dipole.
Finally, hydrocarbons cannot hydrogen bond because the hydrogen (H) is not attached to a N, O, or F.
It is not Van der Waals because this actually includes London as well as dipole-dipole.
Finally, hydrocarbons cannot hydrogen bond because the hydrogen (H) is not attached to a N, O, or F.
Classifying Hydrocarbons
Which two of the following molecules are not hydrocarbons? (More than one answer: check all that apply)
Solution
Hydrocarbons only contain carbon (C) and hydrogen (H). You will learn about molecules containing C, H, and O next year. (Alcohols, Aldehydes, Ketones, Ethers, and Carboxylic Acids).
Classifying Reaction Types
Determine the classification of the reaction:
Solution
C7H16 + 11O2 → 7CO2 + 8H2O
Combustion reactions combine hydrocarbons + oxygen to form carbon dioxide + water.
Physical Property of Hydrocarbons: Boiling Point
Which of the following hydrocarbons will be liquid at SATP (25˚C and 100kPa)? (More than one answer: check all that apply) Solution
| Hydrocarbon | Boiling Point (˚C) |
| CH4 | -164 |
| C3H6 | -42 |
| C4H10 | -1 |
| C5H12 | 36.1 |
| C8H18 | 125 |
CH4, C3H6, and C4H10 have lower boiling points so will be gases at SATP (25˚C and 100kPa).
Physical Property of Hydrocarbons: Boiling Point
C4H10 has a lower boiling point than C14H30 because it is lighter, or in other words, because C4H10 has a lower molecular weight than C14H30.
Physical Property of Hydrocarbons: Boiling Point
The boiling points of five hydrocarbons are given in the table.
| Hydrocarbon | Boiling Point (˚C) |
| C4H10 | -1 |
| C5H12 | 36.1 |
| C8H18 | 125 |
| C10H22 | 174.1 |
| C14H30 | 253 |
Mixing equal parts of C10H22 and C5H12 would result in a hydrocarbon mixture with approximately what boiling point (in ˚C)?
Solution
The boiling point will be approximately between 36.1 ˚C and 174.1 ˚C.
Butane (C4H10) could be separated from the mixture of the five hydrocarbons in the table by bringing the mixture to room temperature (25˚C).
Solution
A 25˚C temperature would cause only butane to become a gas, the rest would be (mostly) in liquid phase (besides a small amount of evaporation of the lighter hydrocarbons). This gas could then be condensed into liquid and collected
Which of the following actions would allow a chemist to separate and collect only C14H30?
Solution
The 200 ˚C temperature would cause the lighter C4-10 hydrocarbons to evaporate into a gas, leaving most of the C14H30 behind as a liquid that can easily be collected.
Describe a process that would allow a chemist to separate and collect only C8H18?
Solution
This is basic fractional distillation.
First, bring the mixture to a temperature lower than C8H18, around 100˚C. This will cause the lighter C4 and C5 to evaporate away into a gas, leaving a liquid mixture of C8, C10, and C14 behind.
Then bring the mixture to a temperature above the boiling point of C8H18, around 150˚C. This would evaporate only the C8H18 into a gas, which could be collected, leaving behind the C10 and C14.
First, bring the mixture to a temperature lower than C8H18, around 100˚C. This will cause the lighter C4 and C5 to evaporate away into a gas, leaving a liquid mixture of C8, C10, and C14 behind.
Then bring the mixture to a temperature above the boiling point of C8H18, around 150˚C. This would evaporate only the C8H18 into a gas, which could be collected, leaving behind the C10 and C14.
Alkanes
Alkanes are hydrocarbons with only single bonds, with a molecular formula with the following ratio of carbons to hydrogens. Which is not an alkane?
Solution
CnH2n + 2
Alkanes all have the molecular formula:
Cn H(2n + 2)
It would be C3H8.
Cn H(2n + 2)
It would be C3H8.
Combustion Reactions
Identify the incomplete combustion reaction.
Solution
Complete combustion creates carbon dioxide (CO2), while incomplete combustion creates carbon monoxide (CO).
Naming Alkane Hydrocarbons
Name the hydrocarbon.
Solution
C7H16
The prefix for 7 is hept-
Heat and Temperature
Heat and temperature are the same.
Solution
Heat describes the moving of energy from one thing to another.
Temperature is a measurement of the thermal energy of something.
Temperature is a measurement of the thermal energy of something.
Specific Heat Capacity
Specific heat capacity is the same for all kinds substances.
Solution
Specific heat capacity is different for all substances.
Substances with higher specific heat capacities can hold more heat for a given amount of mass.
Specific Heat Capacity
The units of specific heat capacity (c) can be J/g˚C or J/kgK.
Solution
Heat Exchange
An increase in heat is indicated as:
Specific Heat Capacity
Specific heat capacity (c) of silver is 0.24 J/g˚C
q = mc∆T100J of heat is lost from a 2.5kg sample of silver with an initial temperature of 10˚C. Find the final temperature.
Solution
T =
Hint
Clear
Info
Incorrect Attempts:
CHECK
˚C
Hint Unavailable
Heat lost is negative, therefore q = -100J
If the same 10˚C block of silver was placed into a 1000kg pool of water at 10˚C, determine the heat (q) of the silver.
Solution
Heat is the movement, or transfer of energy between things with different temperatures. If the temperature is the same (10˚C), then there is no energy transfer. Therefore there is no heat exchange.
Specific Heat Capacity and Latent Heat
When a substance heats up, sometimes it changes from a solid to a liquid, or from a liquid to a gas. These processes use the heat of fusion, and vaporization, respectively.
Latent heat is the heat energy (q) required to alter the molecule proximity when changing the phase of a substance.
Solution
True, for example latent heat of fusion (freezing), or latent heat of vaporization (evaporating)...
The energy to heat an ice cube from -8˚C to water at 30˚C is which of the following? Solution
| Heat Event | Heat (J) |
| Heat ice from -8˚C to 0˚C | x Joules |
| Convert ice from solid to liquid at 0˚C | y Joules |
| Heat water from 0˚C to 30˚C | z Joules |
Thermochemistry: Exothermic and Endothermic
In endothermic reactions
Thermochemistry: Exothermic and Endothermic
The following reaction is exothermic
Solution
Heat is released when the water condenses from higher energy gas to a lower energy liquid. This heat must go somewhere into the surroundings, which makes this condensation reaction exothermic.
Flashcards: Hydrocarbons and Energy
| What is the polarity of hydrocarbons? |
| Non-polar |
| How does the boiling point of a hydrocarbon compare to molecular weight? |
| As molecular weight increases, boiling point increases. (directly proportional) |
| What causes incomplete combustion? |
| A lack of oxygen |
| What is the difference between the products of complete and incomplete combustion? |
| Complete combustion produces carbon dioxide, while incomplete combustion produces a mixture of carbon monoxide and carbon dioxide. |
| What is the difference between heat and temperature? |
| Heat describes the moving of energy from one thing to another. Temperature is a measurement of the thermal energy of something. |
| What is specific heat? |
| The heat (energy) required to raise the temperature of a given material by a certain amount |
| What does the symbol, q represent? |
| Heat (energy) |
| What are the S.I. units of heat? |
| Joules (J) |
| What is latent heat? |
| Latent heat is the heat energy (q) required to alter the molecule proximity when changing the phase of a substance. |
| Can a liquid evaporate when it is below its boiling point? |
| Yes |
| What is the difference between evaporation and boiling? |
| Evaporation can occur below the boiling point of a liquid, while boiling occurs at or above the boiling point. |
| Explain the change in temperature of the system and surroundings in an endothermic process |
| In an endothermic process the system absorbs energy from the surroundings. Therefore the temperature of the system increases and the temperature of the surroundings decrease. |
| Compare the kinetic energy of the products and reactants in an exothermic reaction |
| The kinetic energy of the products is higher than the reactants |
| Compare the potential energy of the products and reactants in an endothermic reaction |
| The potential energy of the reactants is higher than the products |
